Product Information
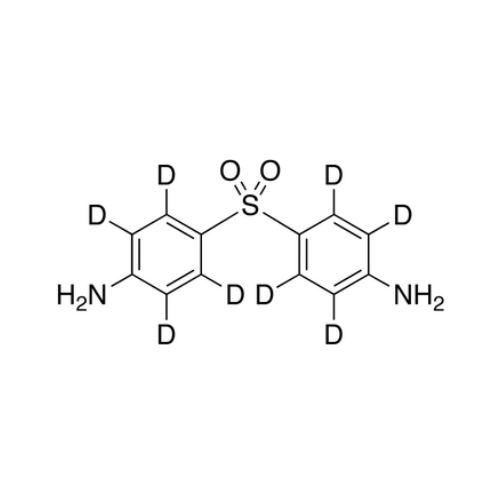
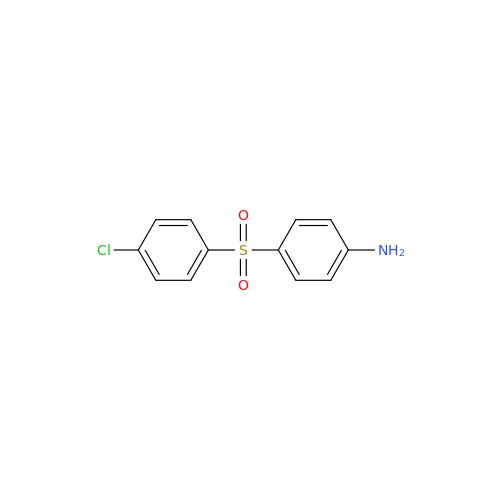
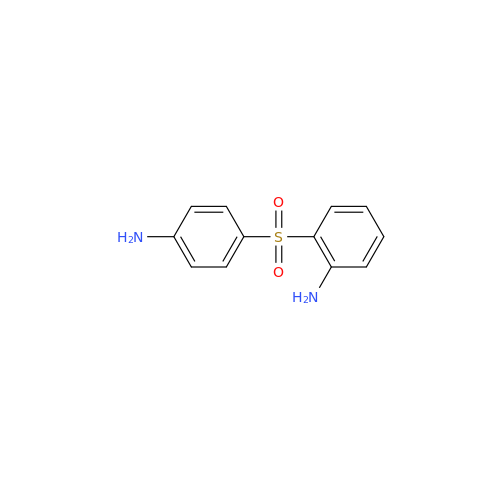
Dapsone EP Impurity D
|
Chemical Name: Dapsone EP Impurity D
Synonym: 2,4'-Diaminodiphenyl Sulphone; 2-((4-Aminophenyl)sulfonyl)aniline; 4,2′-Bis(aminophenyl) Sulfone; 2-(4-Aminophenyl)sulfonylaniline; 2,4′-Diaminophenyl Sulfone; 2,4′-Diaminodiphenyl Sulfone| Enter Batch Number | |||