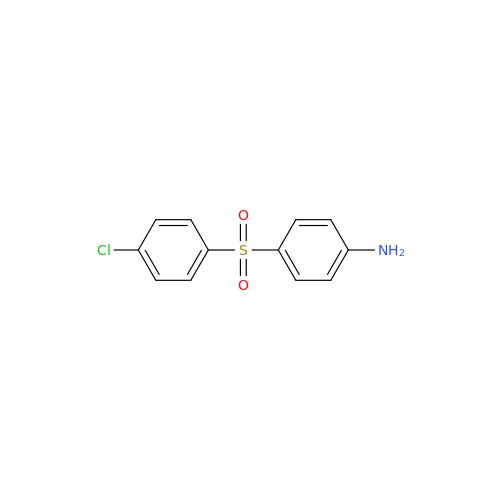
Product Information
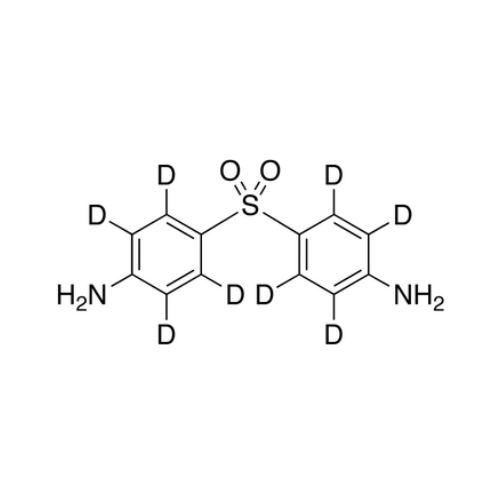
Dapsone EP Impurity A
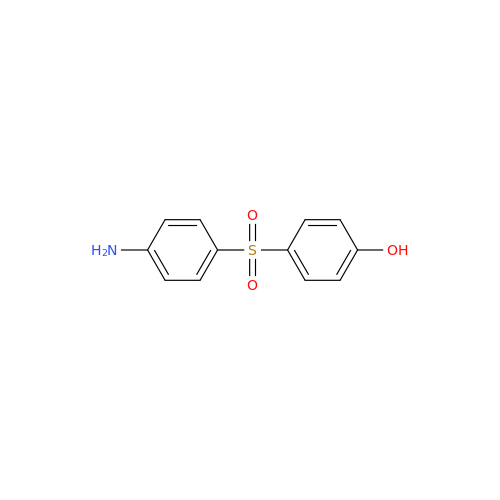
|
Chemical Name: Dapsone EP Impurity A
Synonym: 4-Amino-4'-hydroxydiphenyl Sulphone; 4-[(4-Aminophenyl)sulfonyl]phenol; p-Sulfanilylphenol; 4-(4-Aminobenzene-1-sulfonyl)phenol; 4-((4-Aminophenyl)sulfonyl)phenol| Enter Batch Number | |||