Product Information
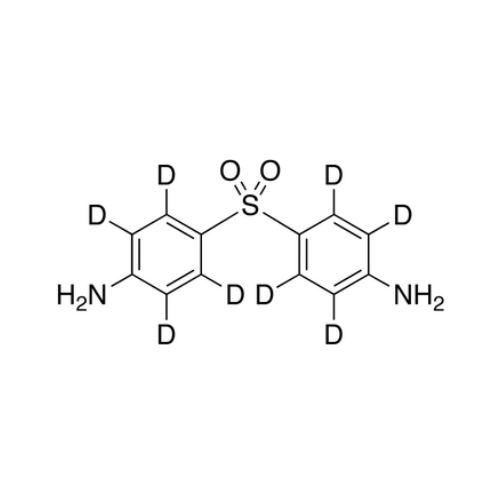
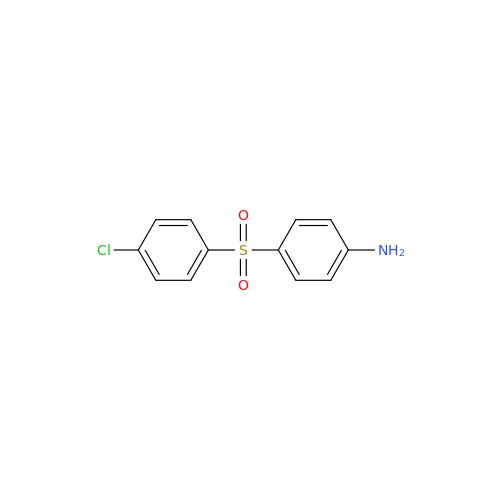
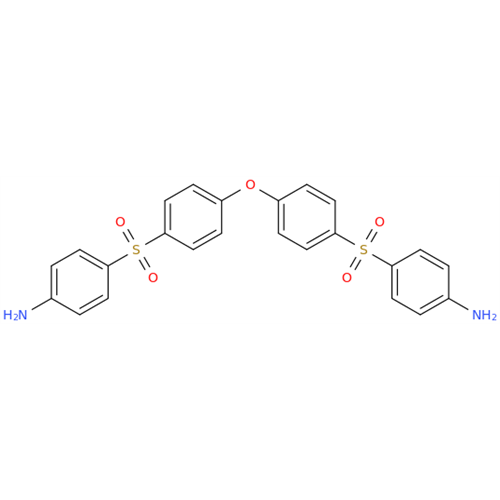
Dapsone EP Impurity C
|
Chemical Name: Dapsone EP Impurity C
Synonym: Dapsone O-Dimer Impurity; 4,4′-[Oxybis[(4,1-phenylene)sulfonyl]]dianiline; 4,4'-(Oxybis(4,1-phenylenesulfonyl))dianiline; 4,4′-[Oxybis[(4,1-phenylene)sulfonyl]]dianiline| Enter Batch Number | |||