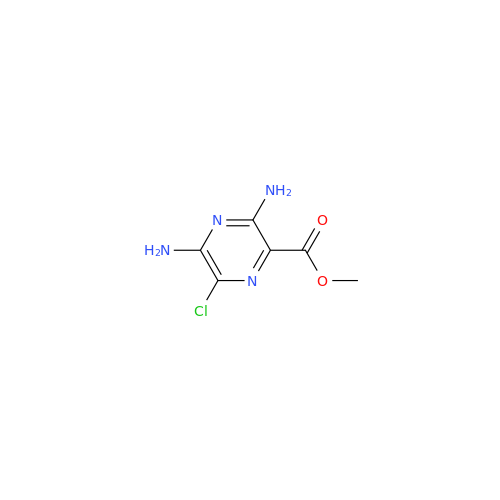
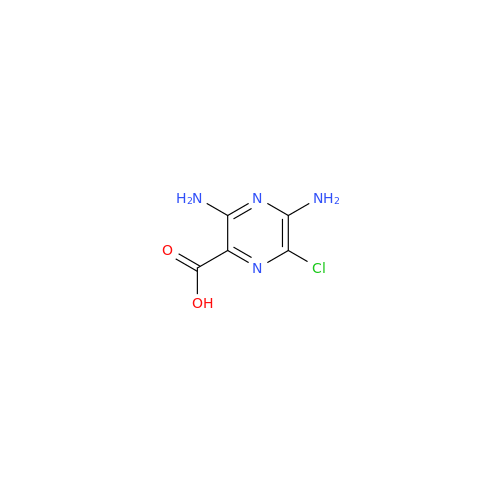
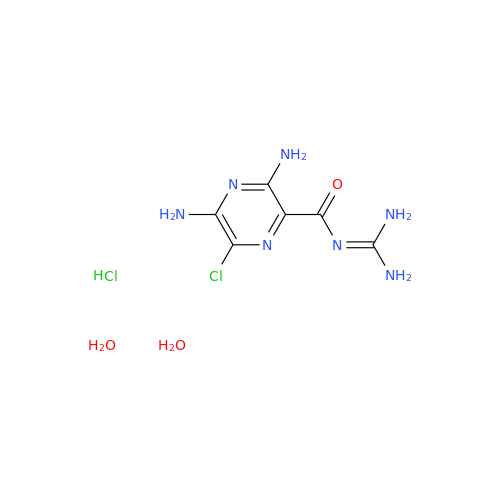
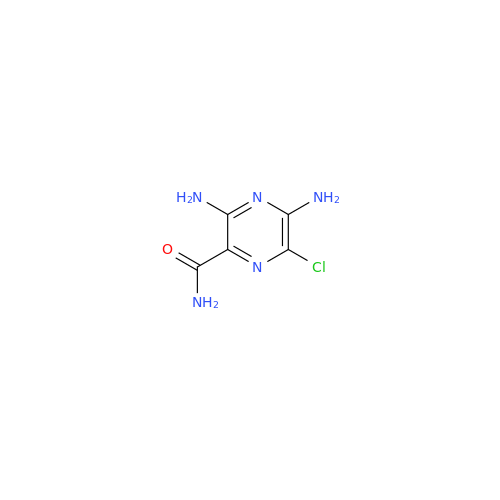
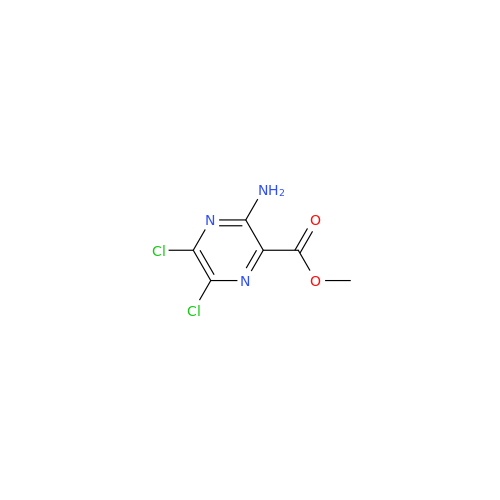
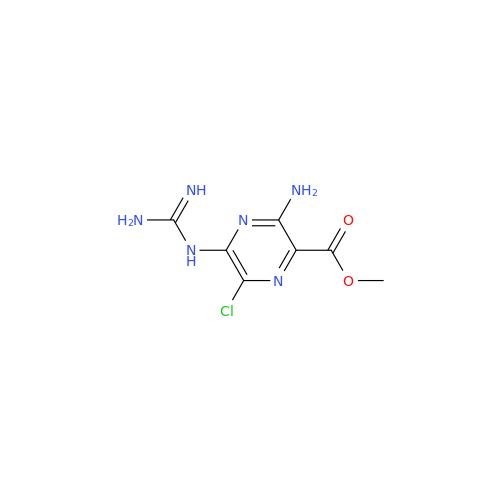
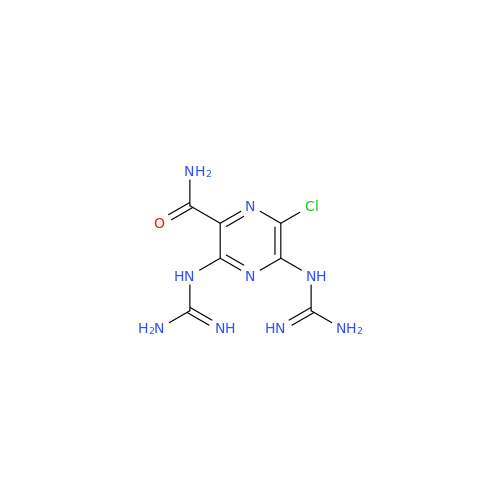
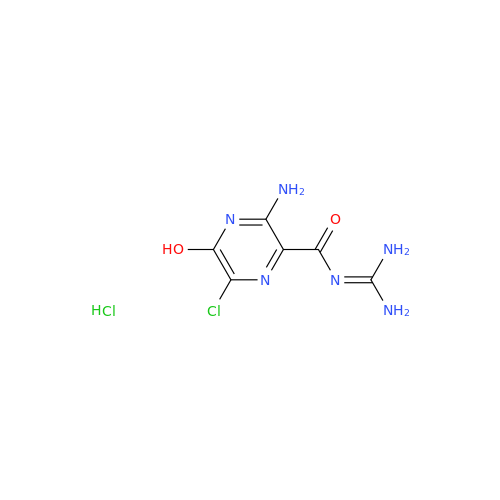
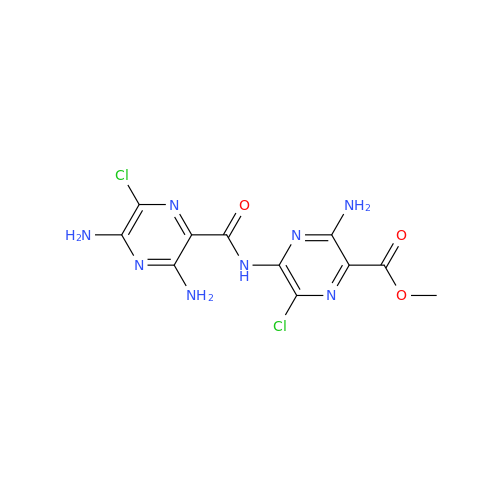
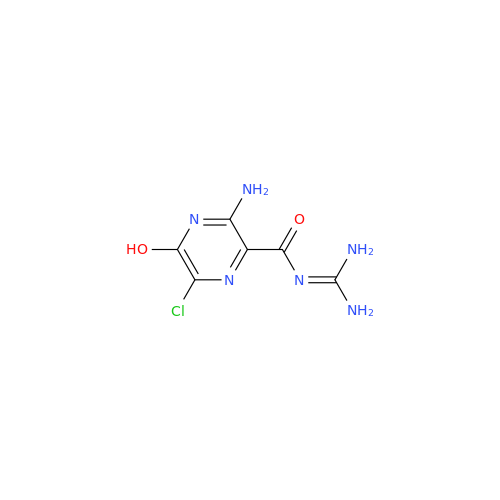
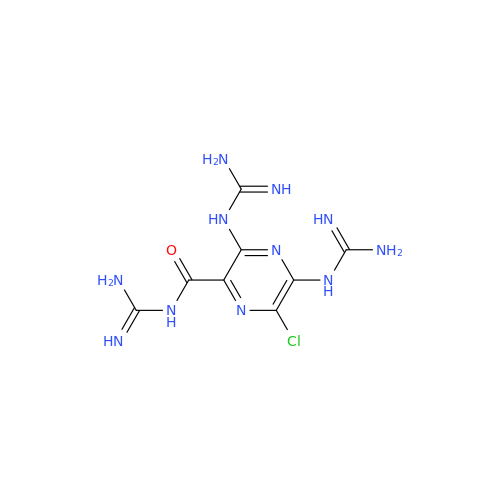
Product Information
Amiloride Impurity 2
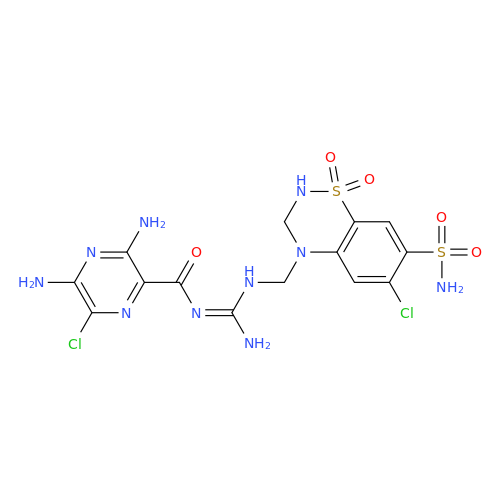
|
Chemical Name: Amiloride Impurity 2
Synonym: (Z)-3,5-Diamino-N-(amino(((6-chloro-1,1-dioxido-7-sulfamoyl-2,3-dihydro-4H-benzo[e][1,2,4]thiadiazin-4-yl)methyl)amino)methylene)-6-chloropyrazine-2-carboxamide| Enter Batch Number | |||