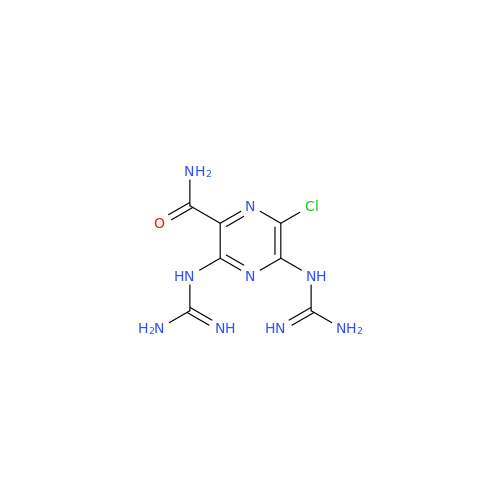
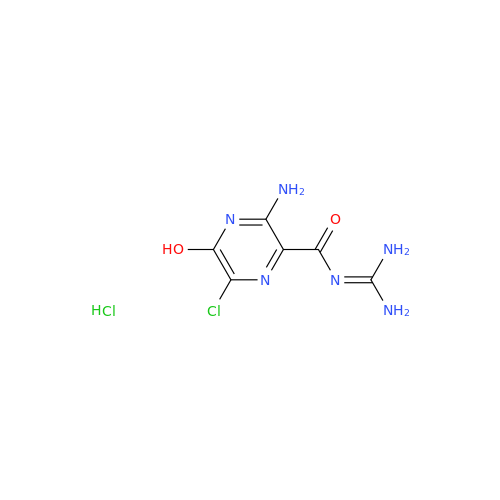
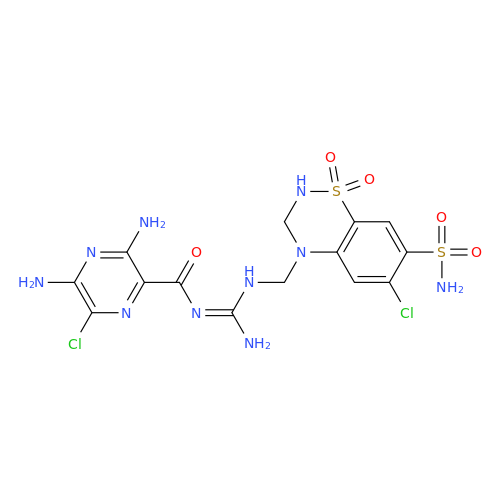
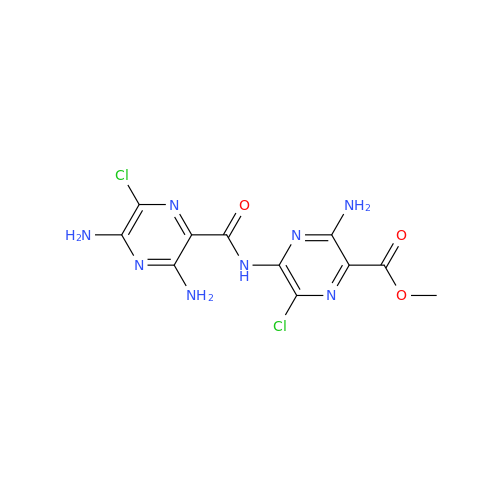
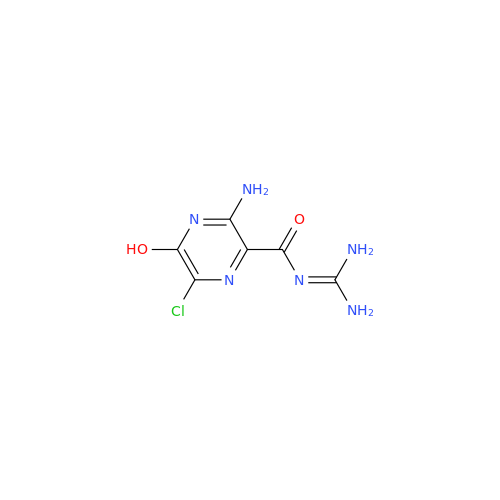
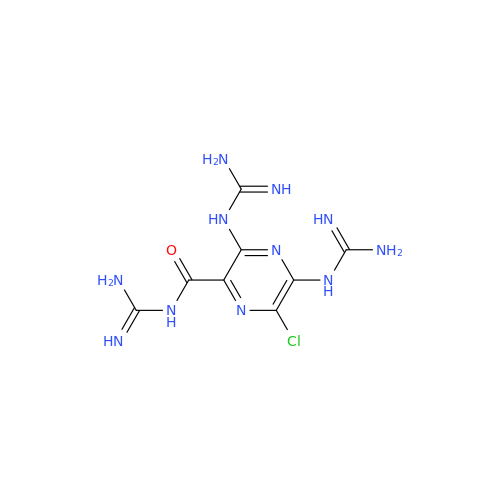
Product Information
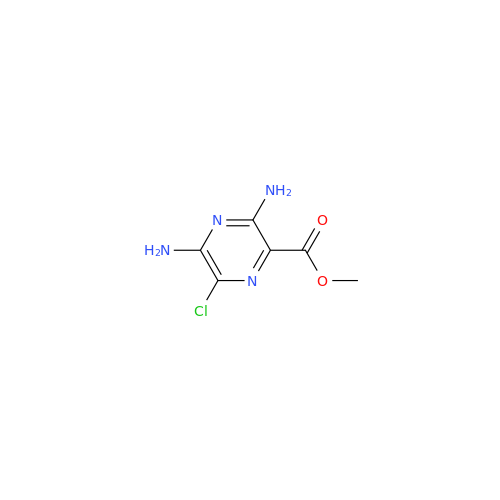
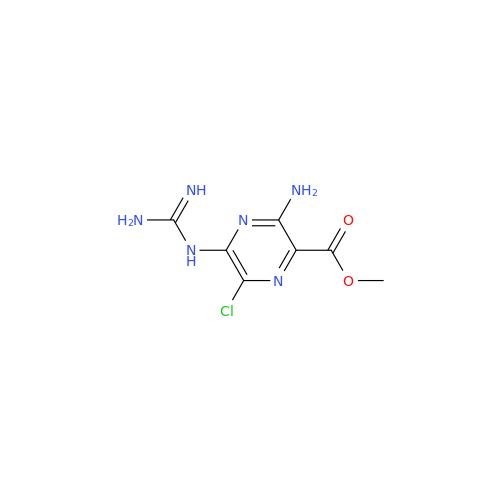
Amiloride EP Impurity B
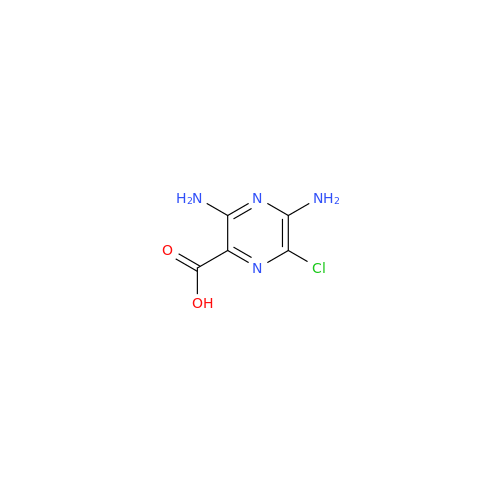
|
Chemical Name: Amiloride EP Impurity B
Synonym: 3,5-Diamino-6-chloro-2-pyrazinecarboxylic Acid; 3,5-Diamino-6-chloropyrazinecarbonic Acid| Enter Batch Number | |||