Product Information
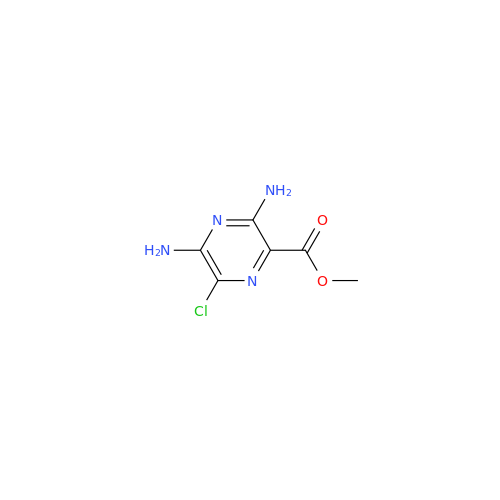
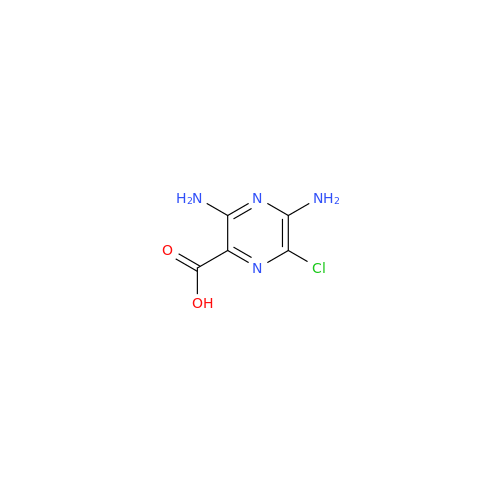
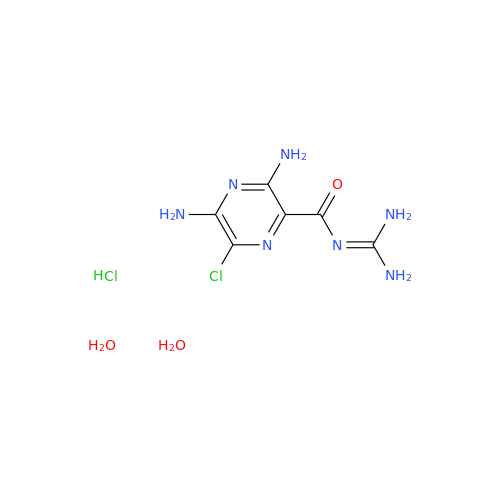
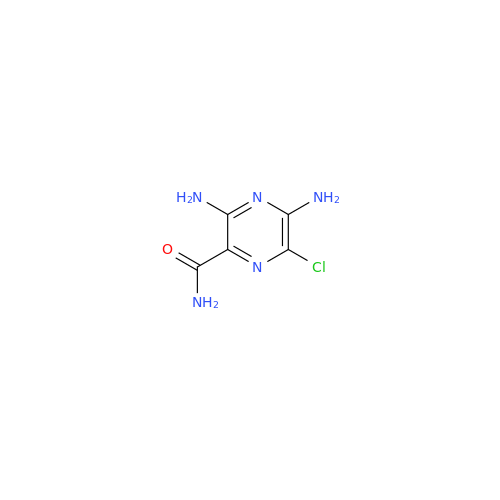
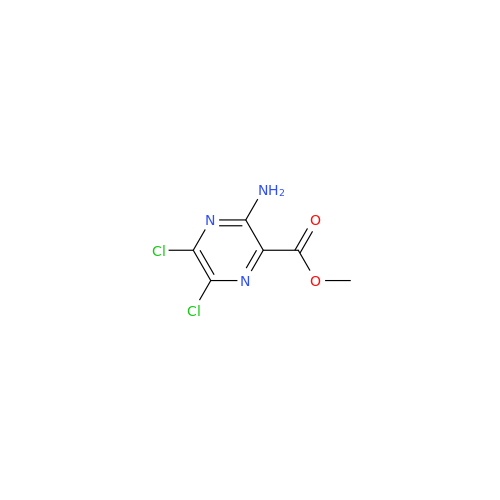
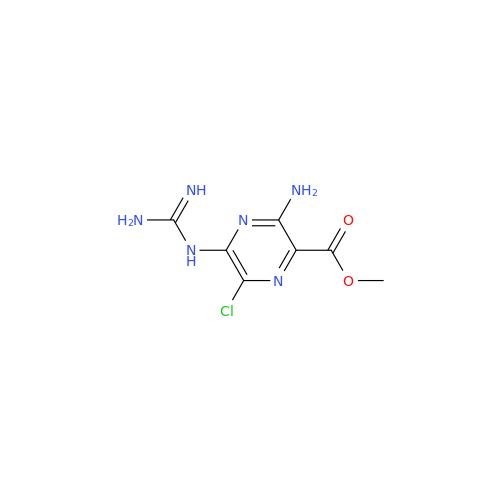
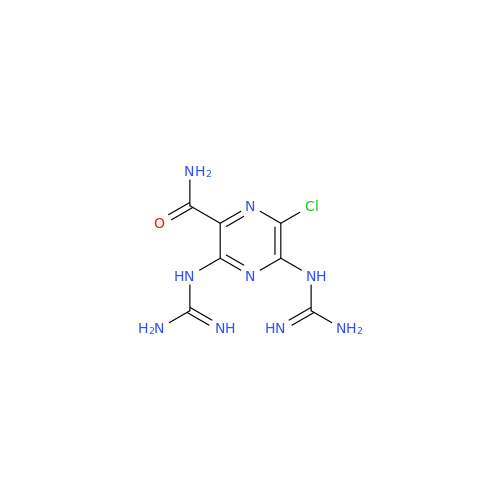
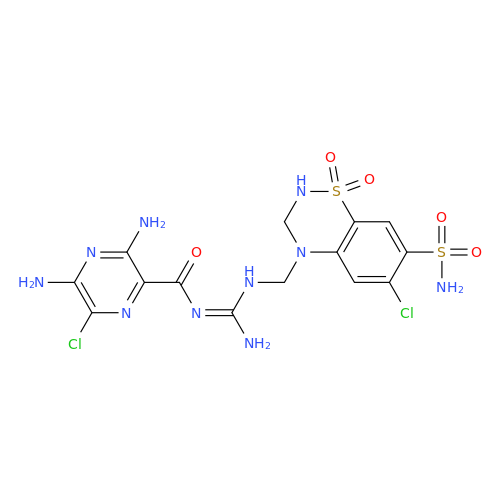
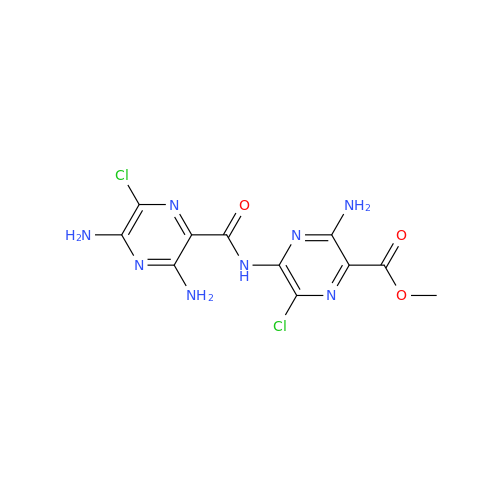
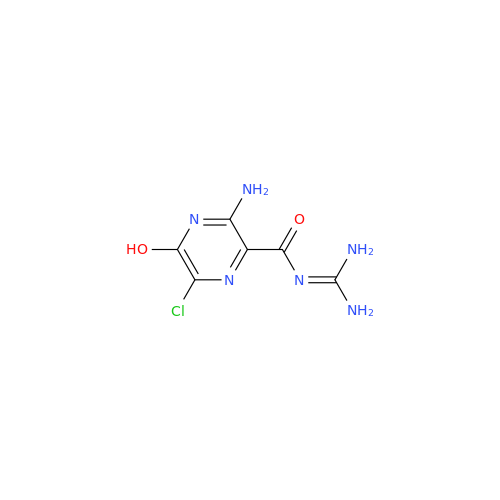
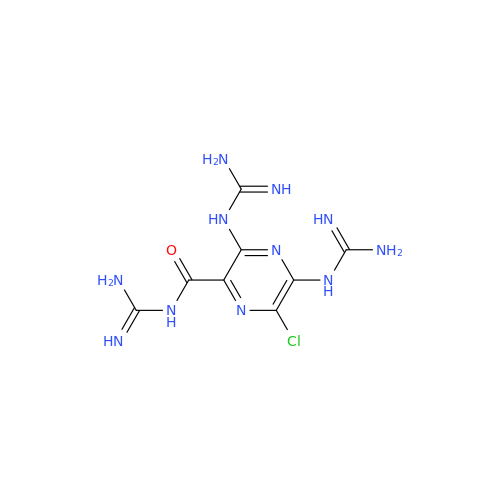
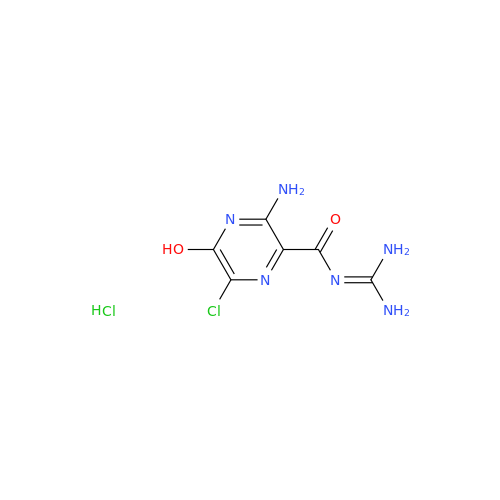
Amiloride EP Impurity C (HCl salt)
|
Chemical Name: Amiloride EP Impurity C (HCl salt)
Synonym: 5-Hydroxypyrazine-2-carboxamide Hydrochloride, 3-amino-6-chloro-N-(diaminomethylene)-| Enter Batch Number | |||