Product Information
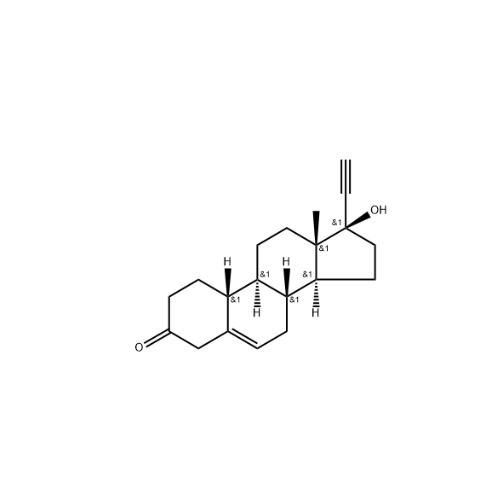
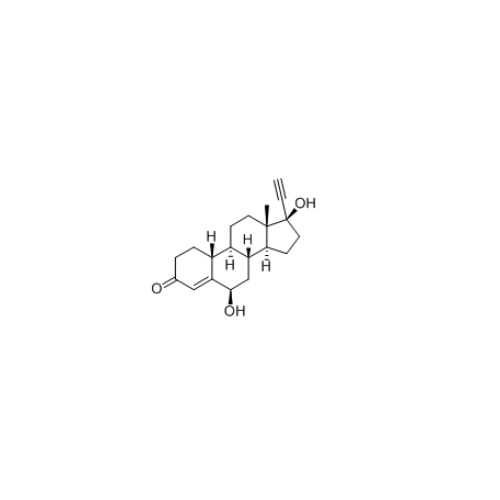
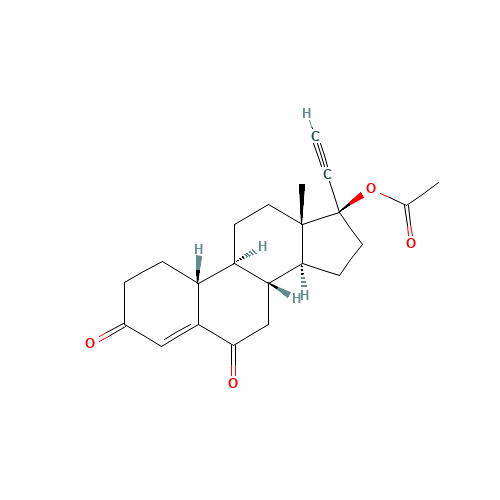
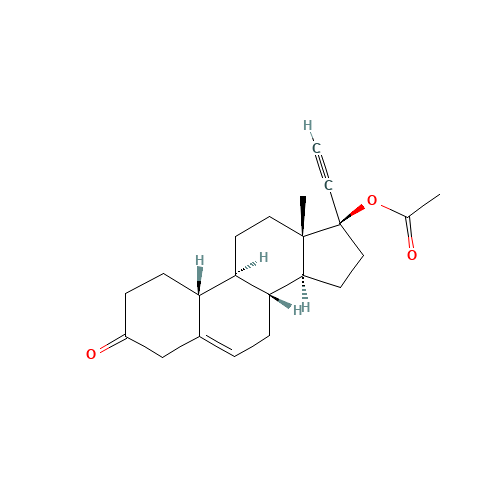
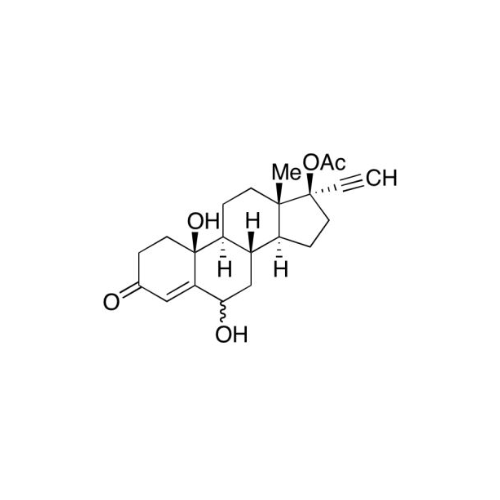
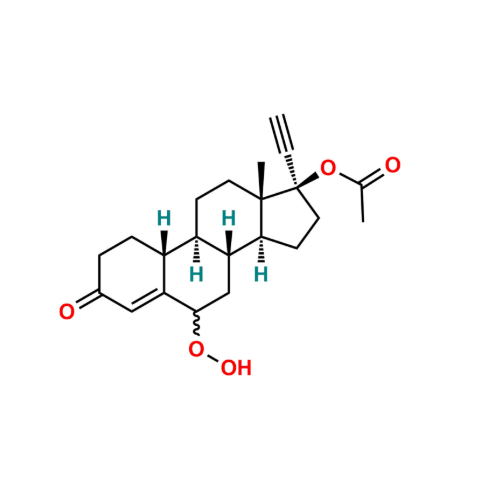
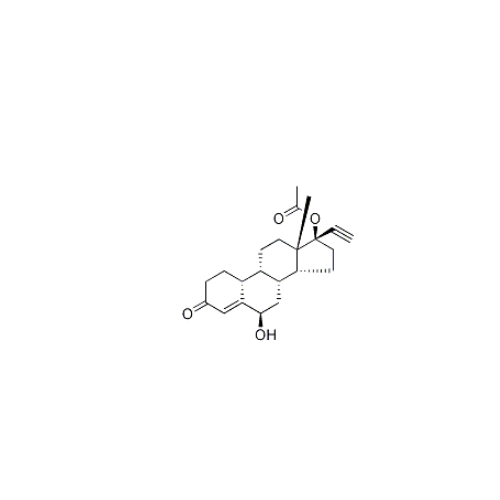
Norethindrone Acetate 6-alpha-Hydroxy Impurity
|
Chemical Name: Norethindrone Acetate 6-alpha-Hydroxy Impurity
Synonym: 6α,17-Dihydroxy-19-nor-17α-pregn-4-en-20-yn-3-one 17-acetate| Enter Batch Number | |||