Product Information
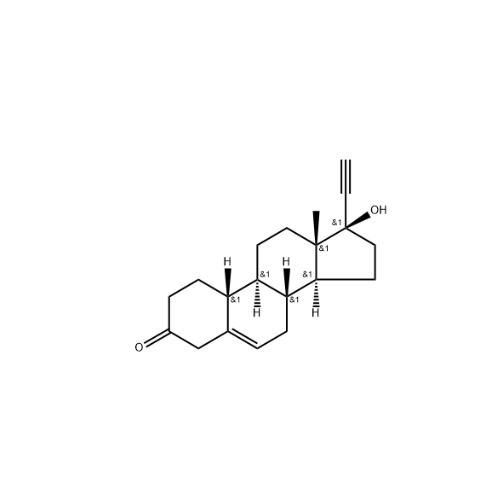
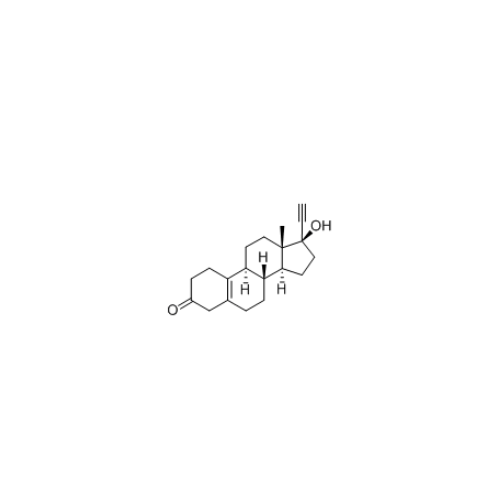
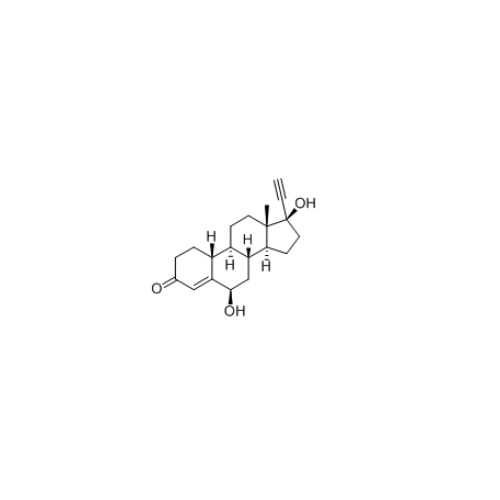
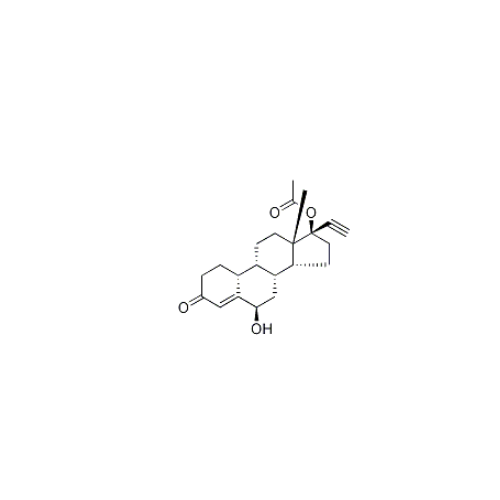
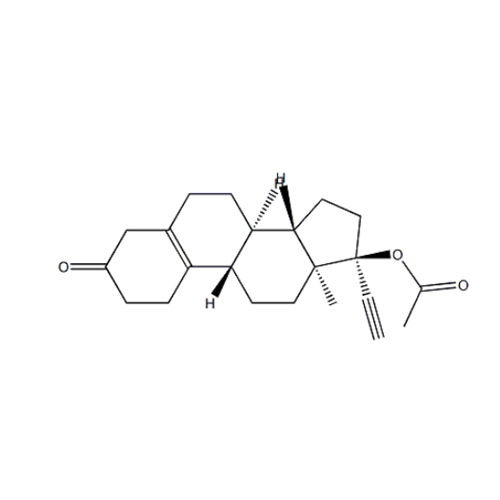
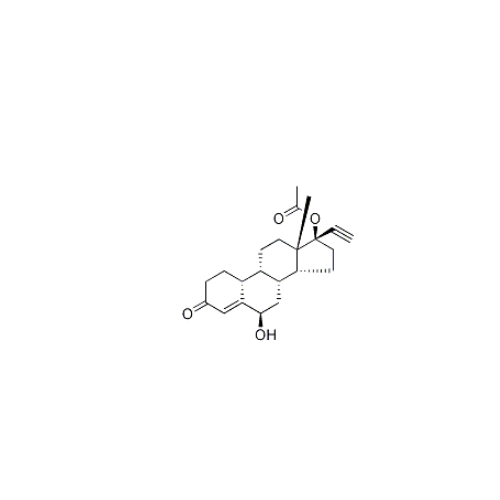
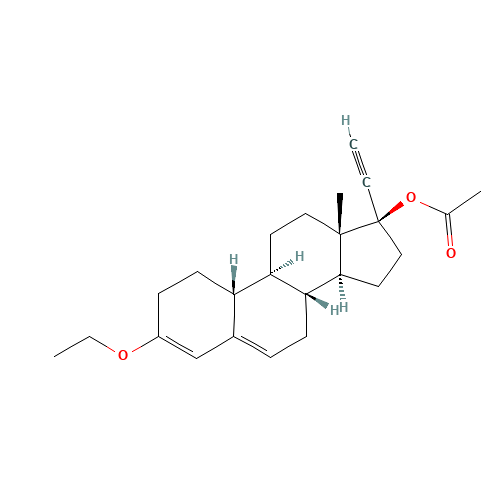
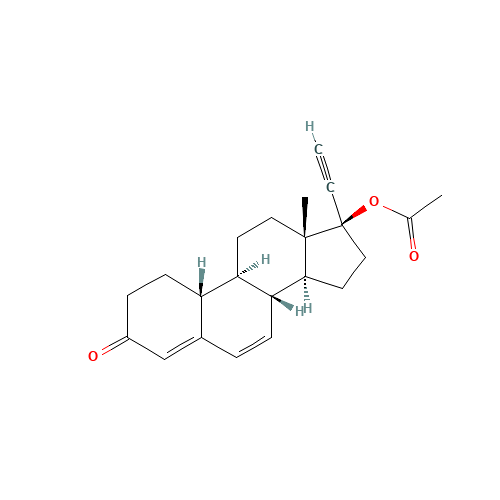
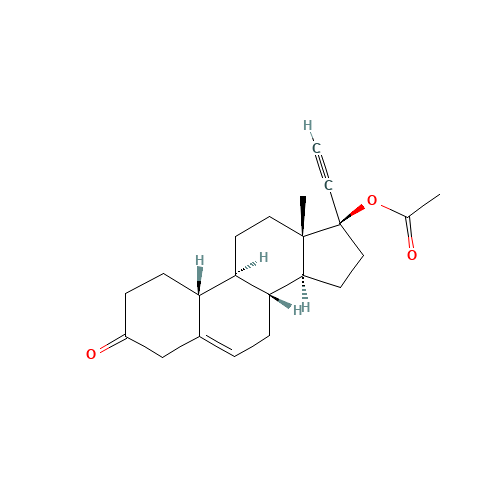
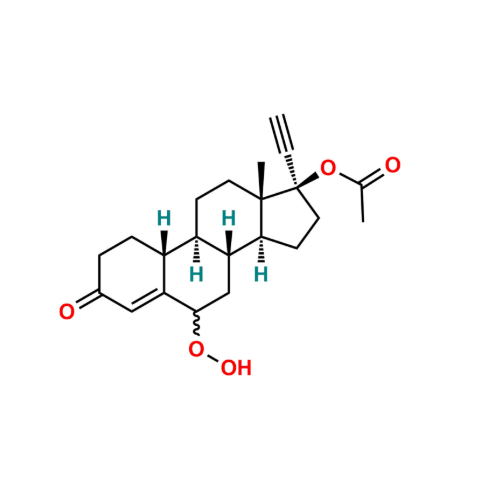
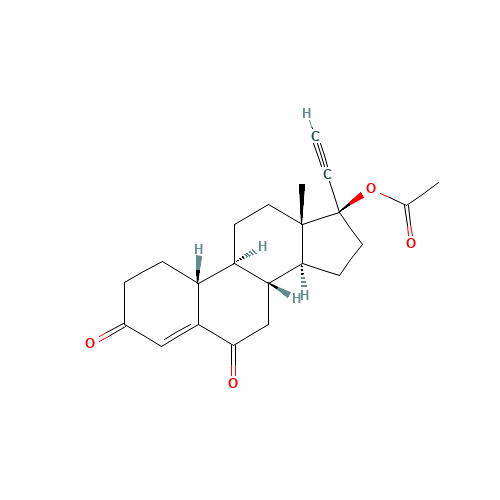
Norethindrone Acetate EP Impurity G
|
Chemical Name: Norethindrone Acetate EP Impurity G
Synonym: Norethindrone Acetate 6-Oxo Impurity ; 6-Ketonorethindrone Acetate| Enter Batch Number | |||