Product Information
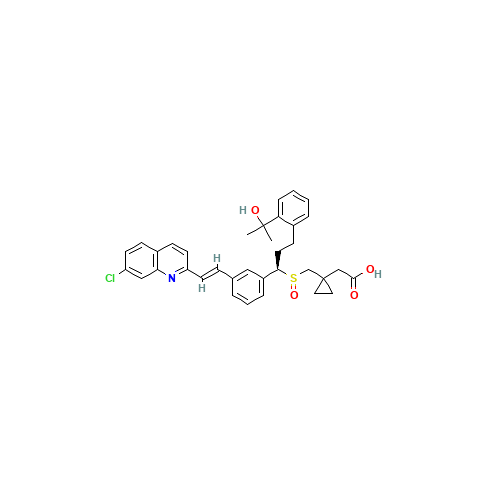
Montelukast Sulfoxide (USP)
|
Chemical Name: Montelukast Sulfoxide (USP)
Synonym:| Enter Batch Number | |||

|
Chemical Name: Montelukast Sulfoxide (USP)
Synonym:| Enter Batch Number | |||