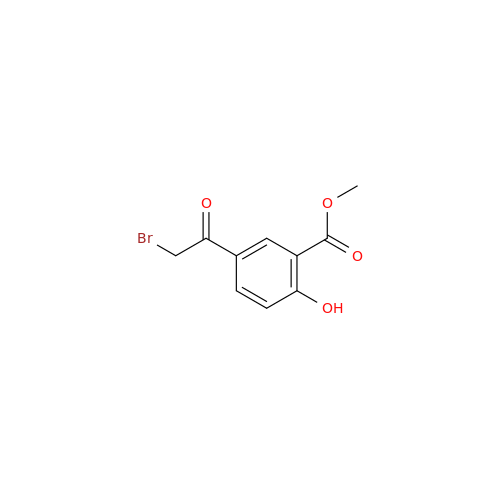
Product Information
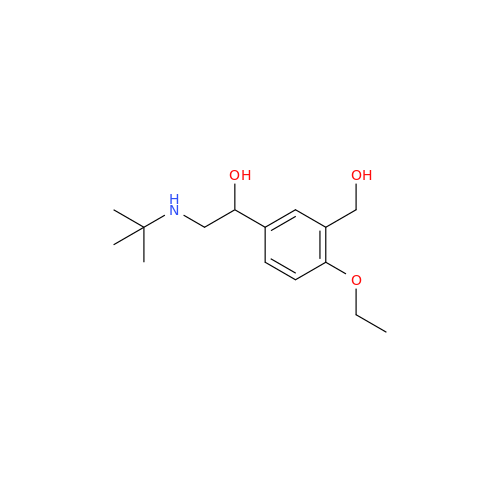
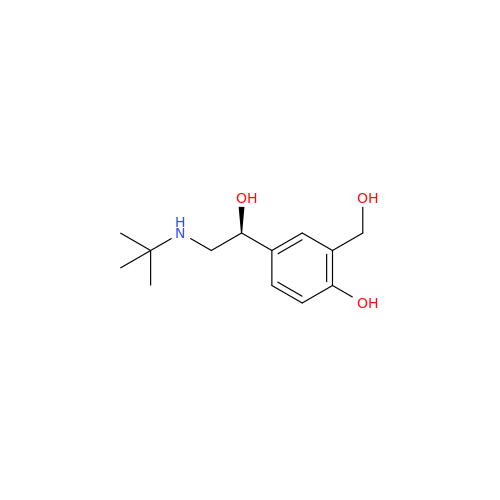
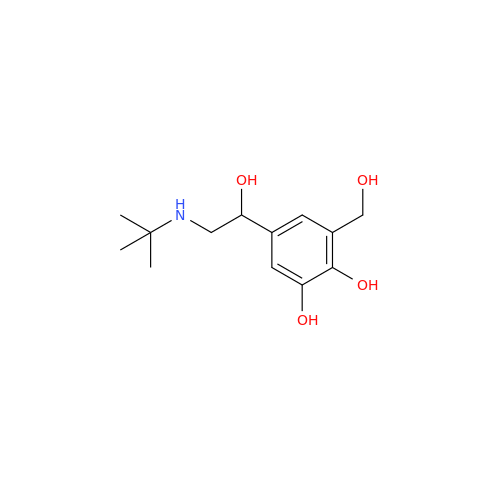
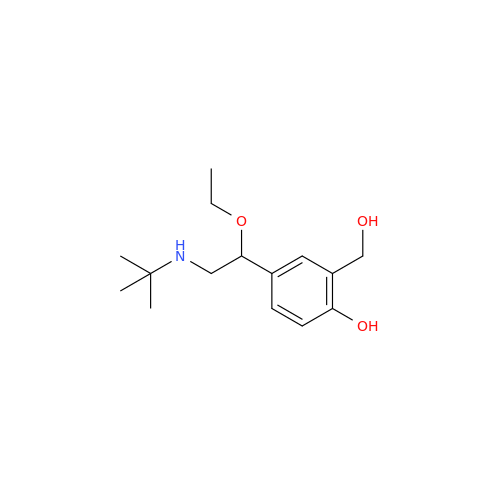
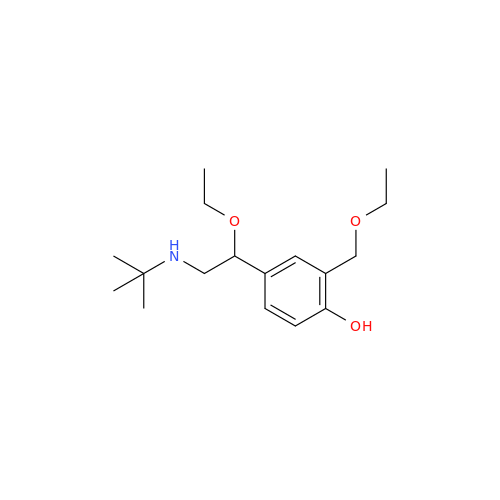
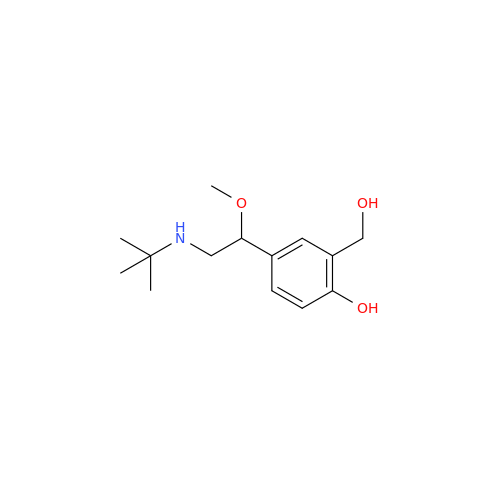
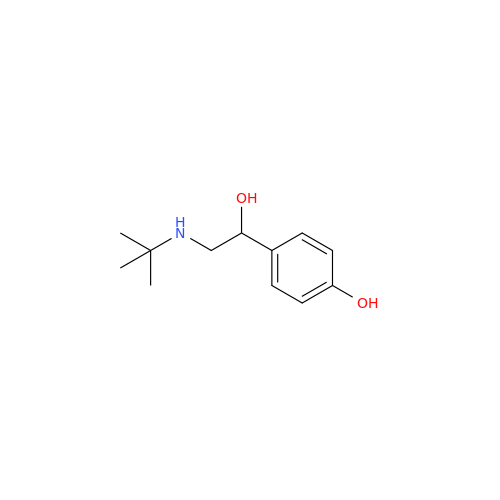
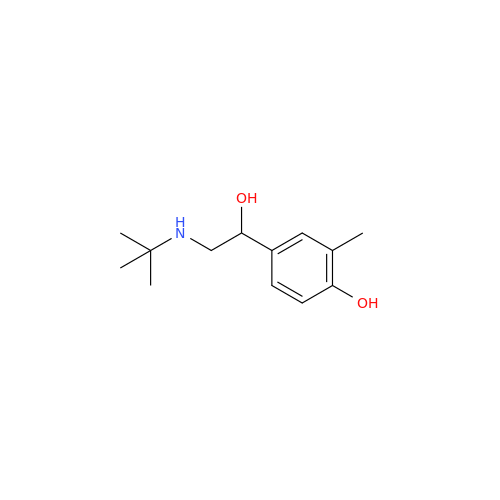
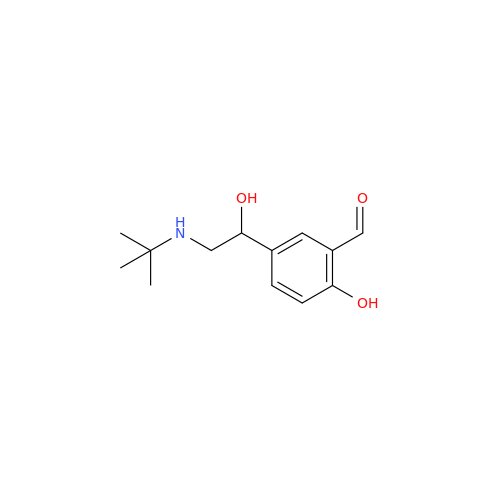
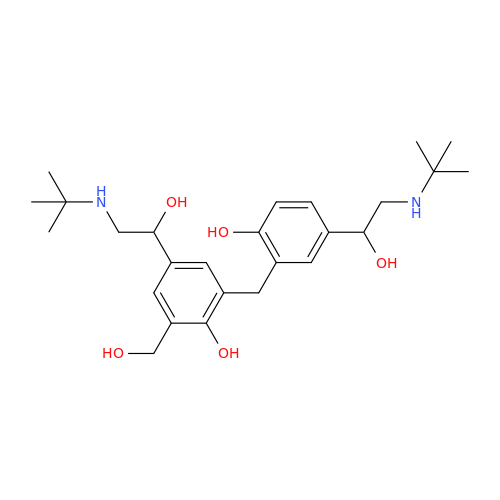
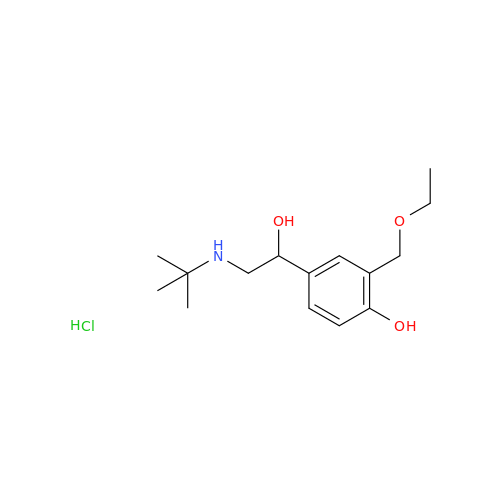
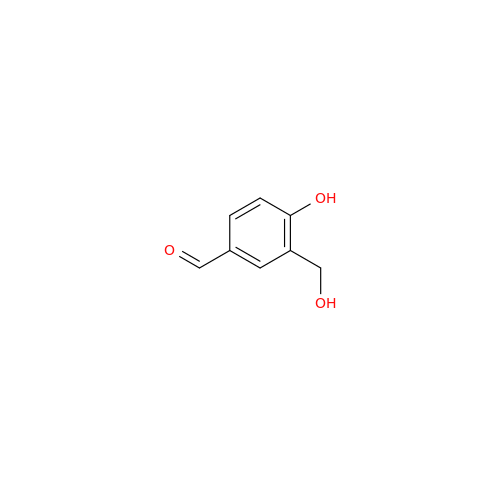
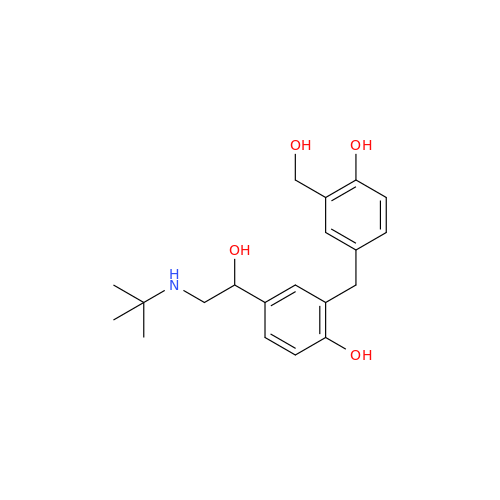
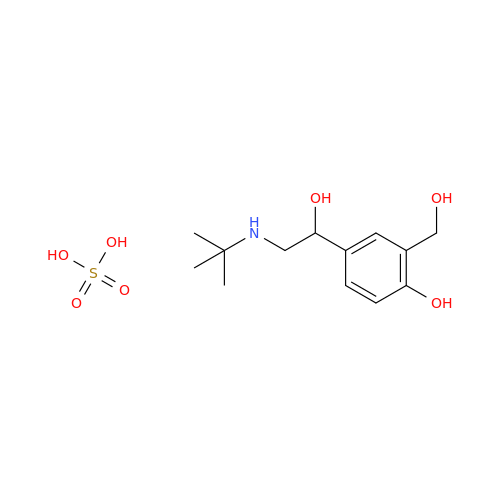
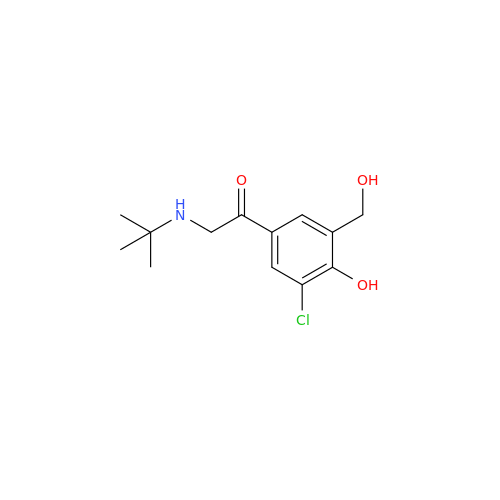
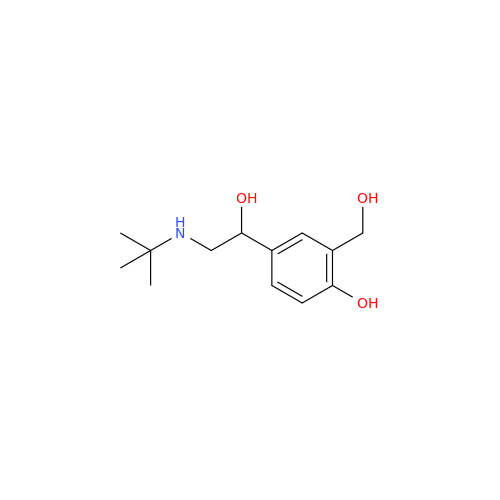
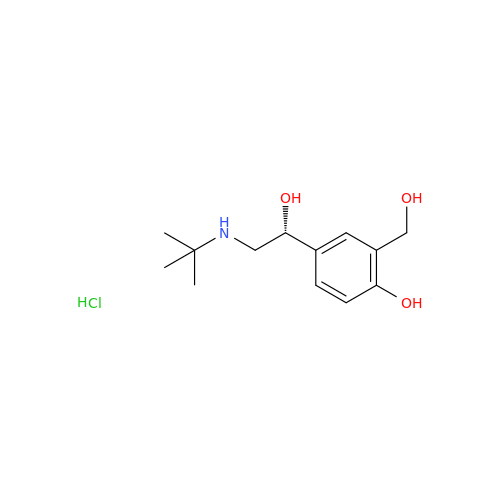
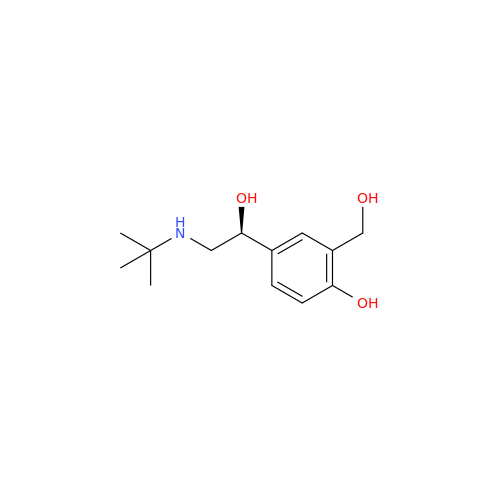
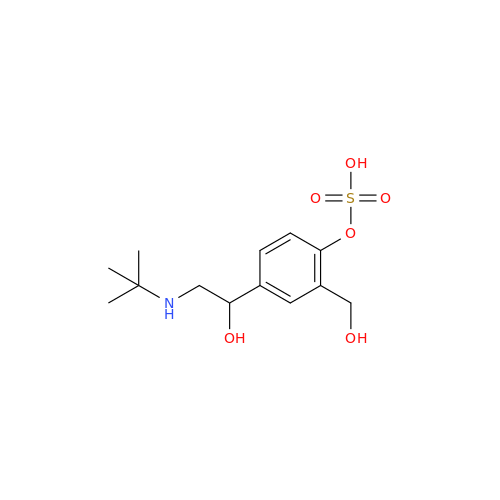
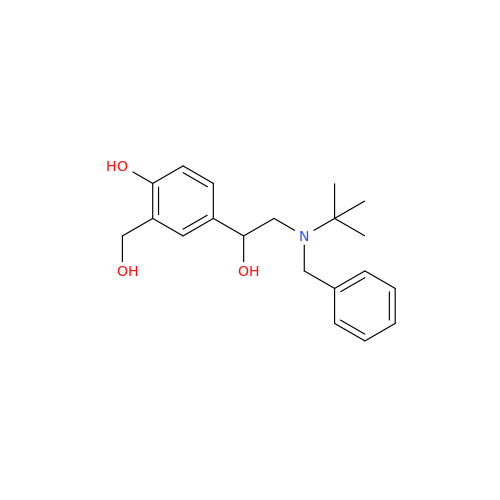
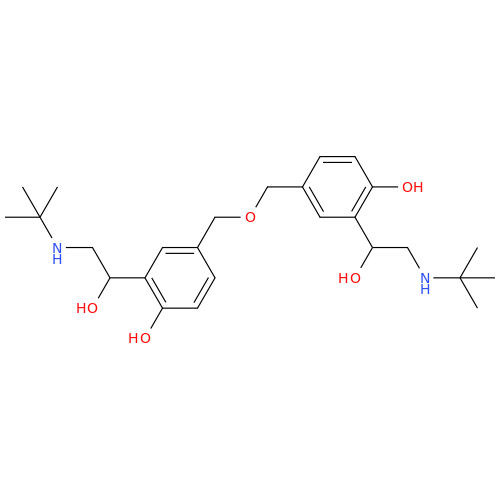
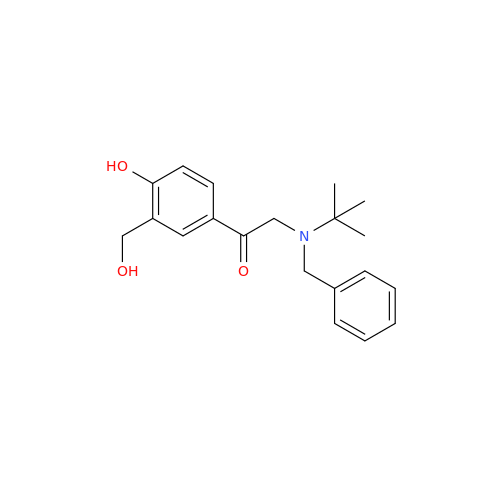
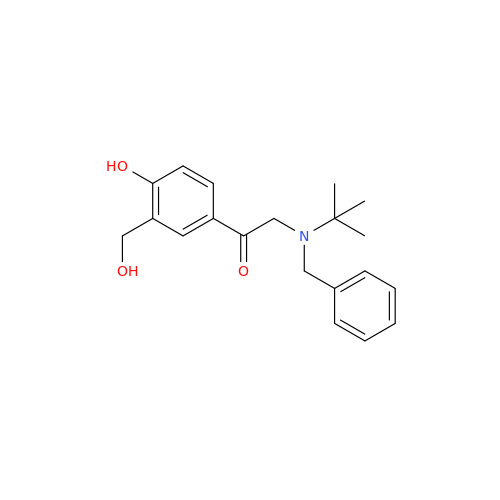
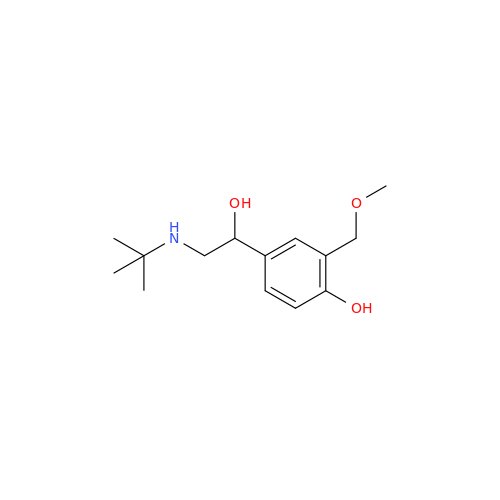
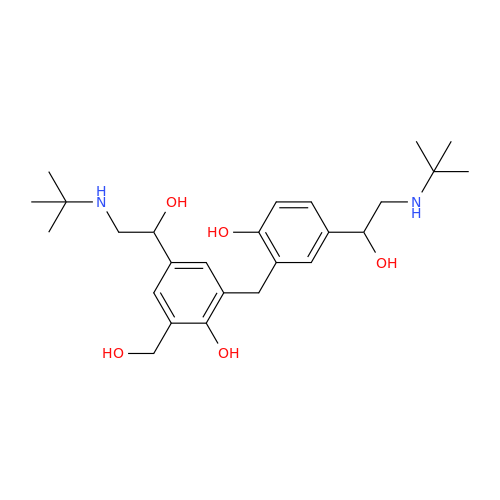
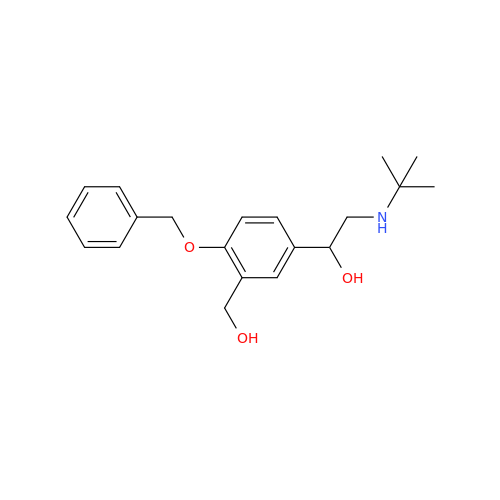
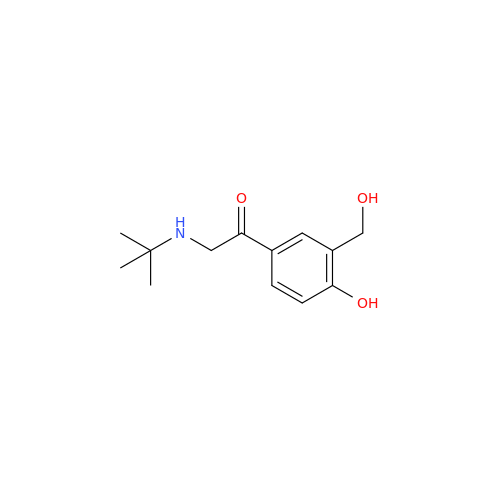
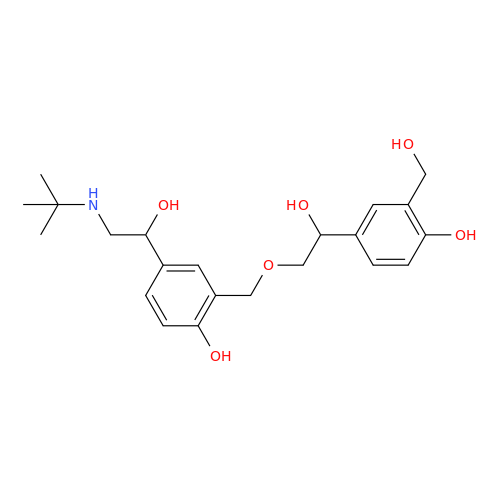
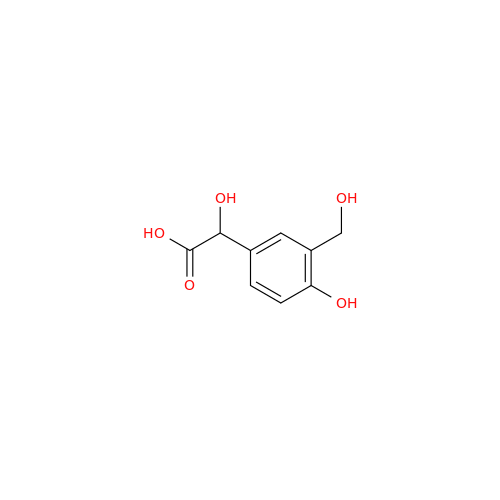
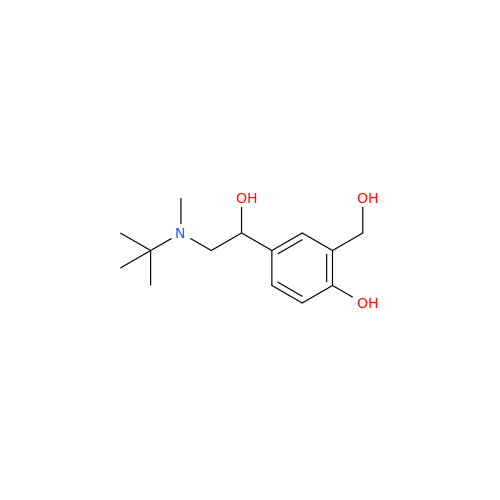
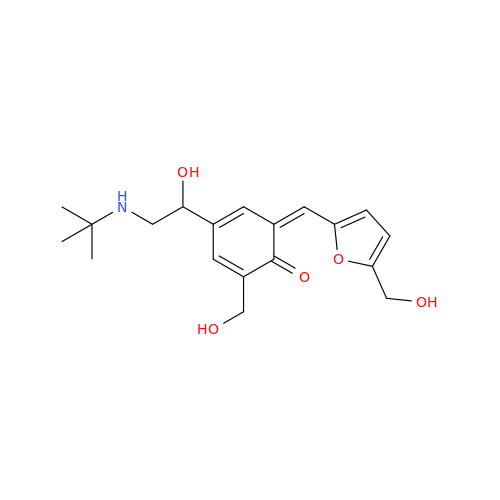
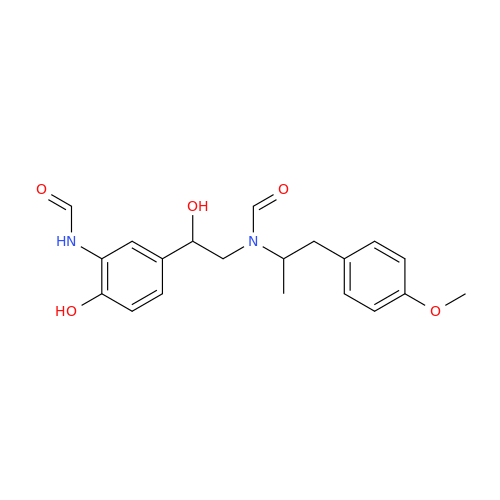
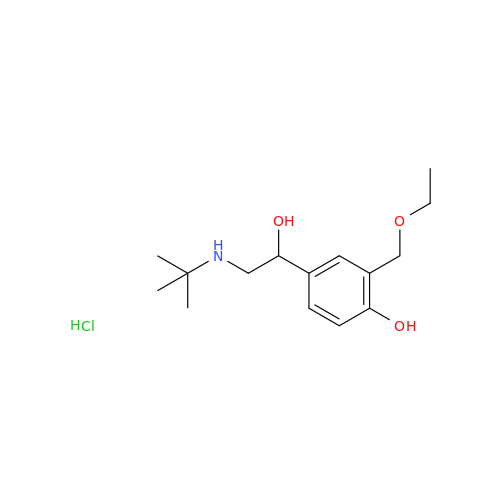
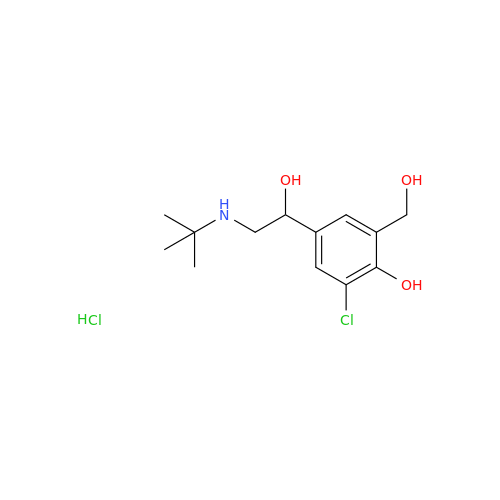
Salbutamol EP Impurity L Hydrochloride
|
Chemical Name: Salbutamol EP Impurity L Hydrochloride
Synonym: 4-[(1RS)-2-(tert-butylamino)-1-hydroxyethyl]-2-chloro-6-(hydroxymethyl)phenol hydrochloride| Enter Batch Number | |||