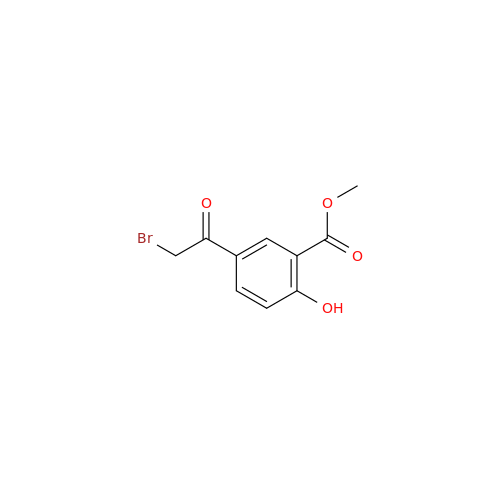
Product Information
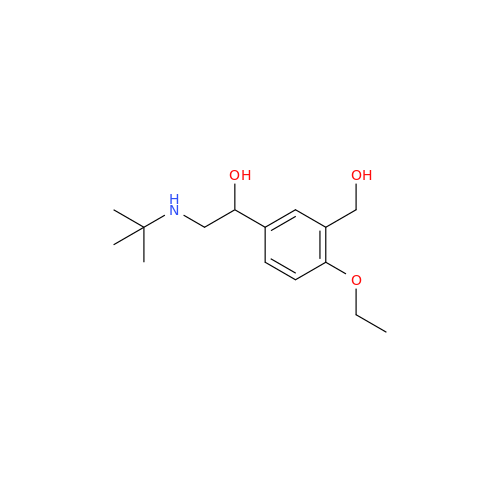
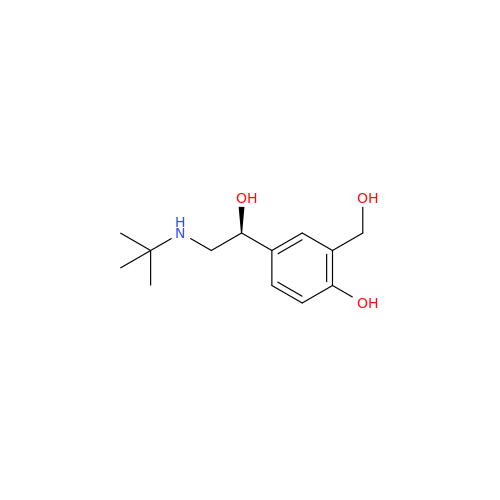
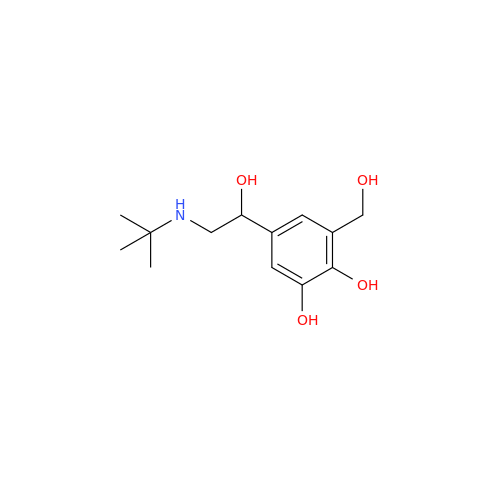
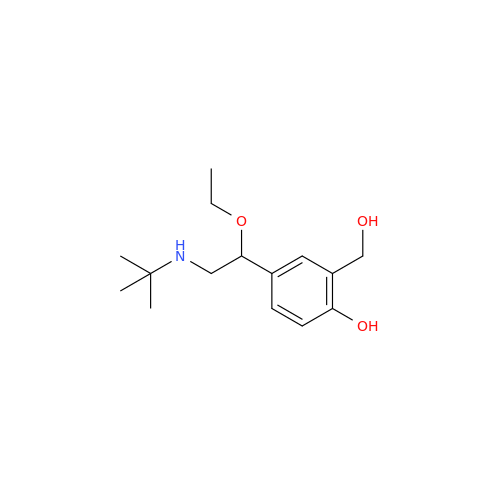
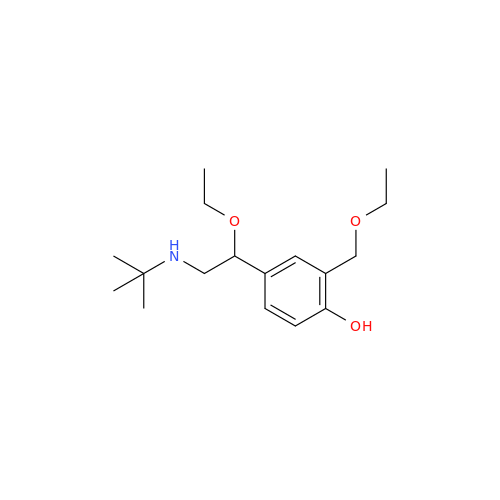
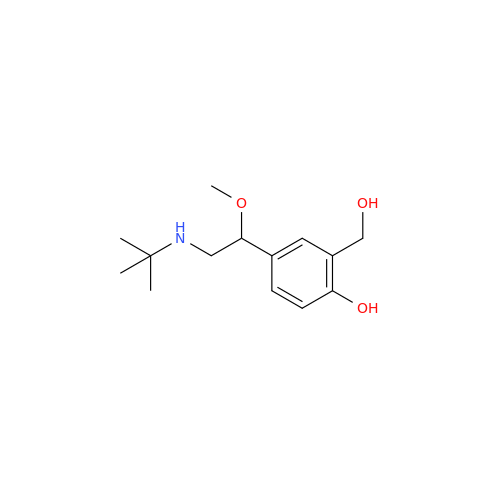
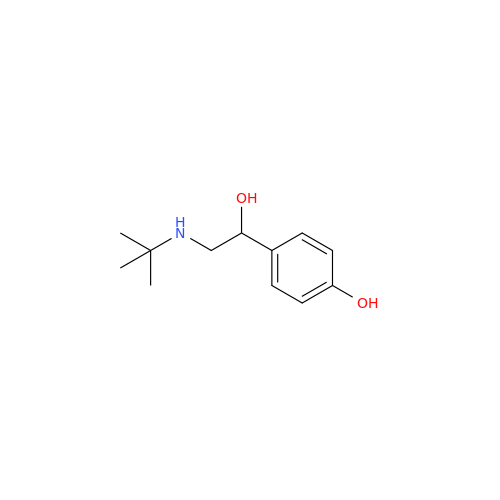
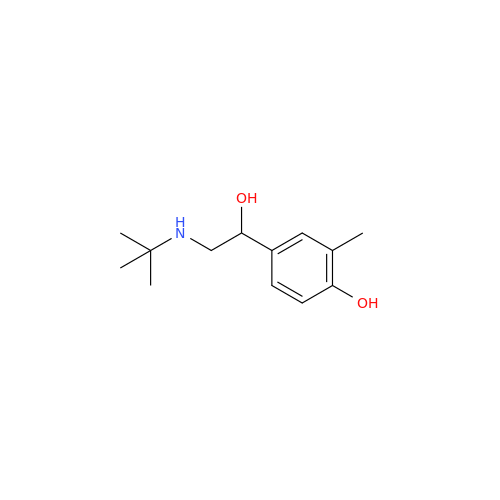
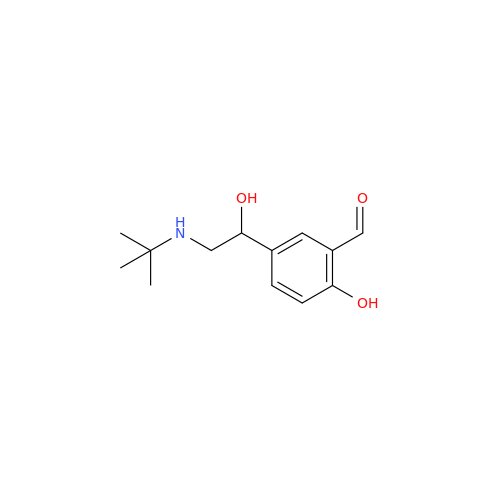
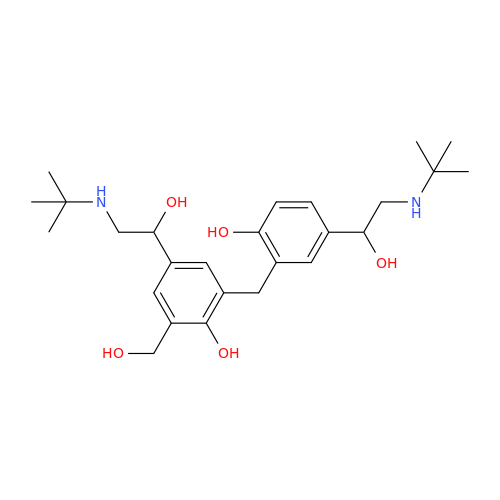
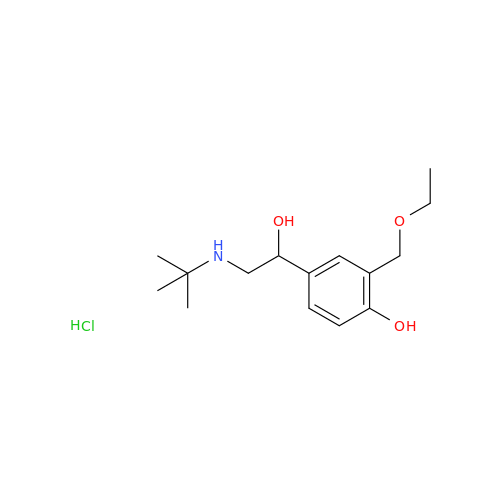
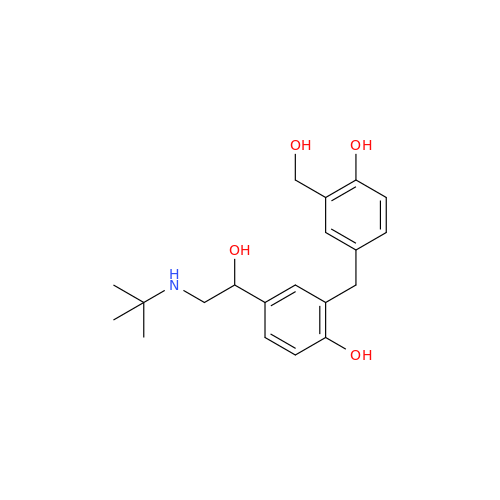
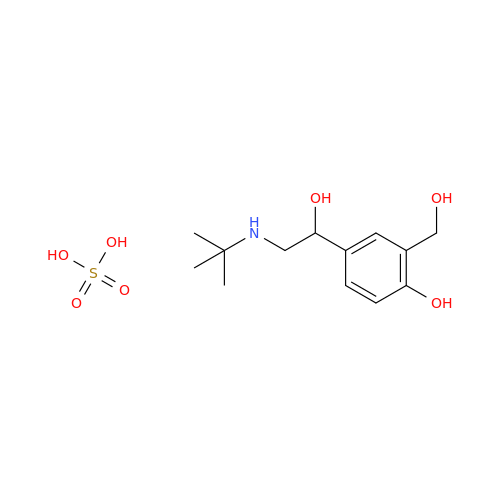
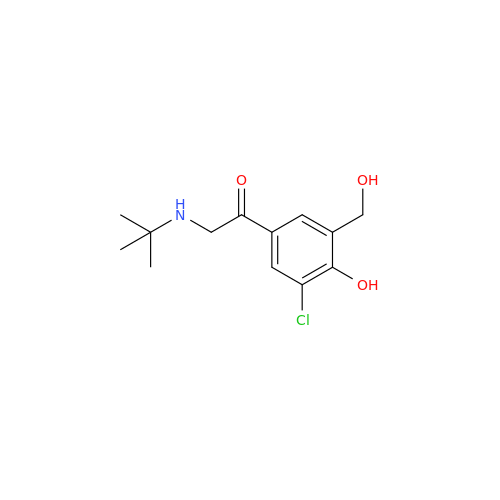
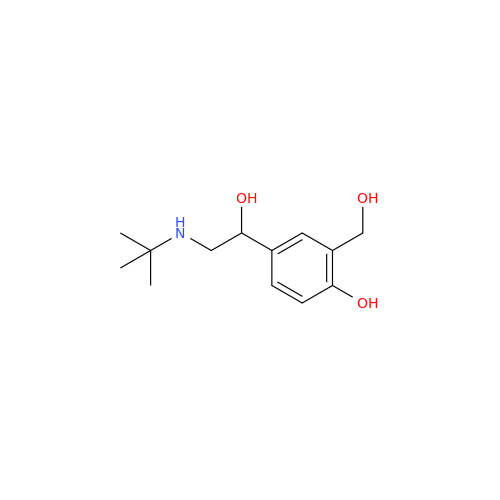
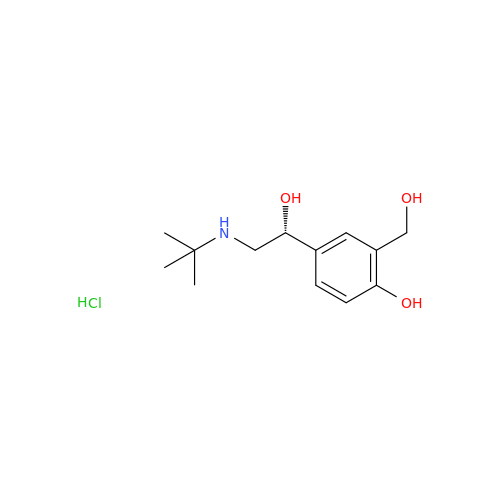
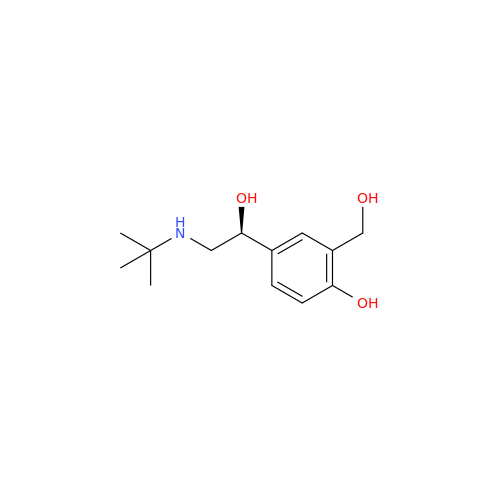
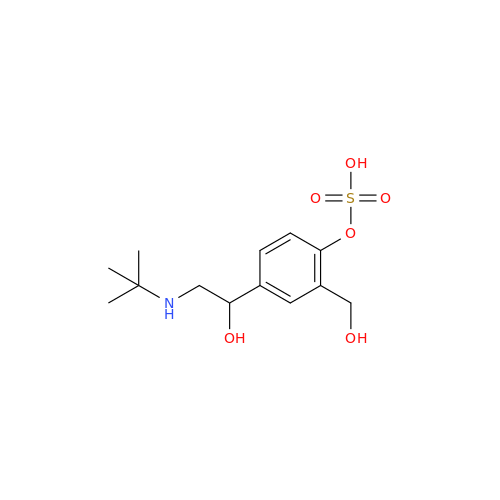
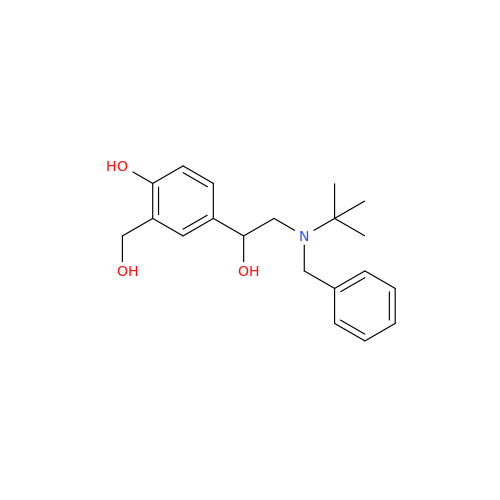
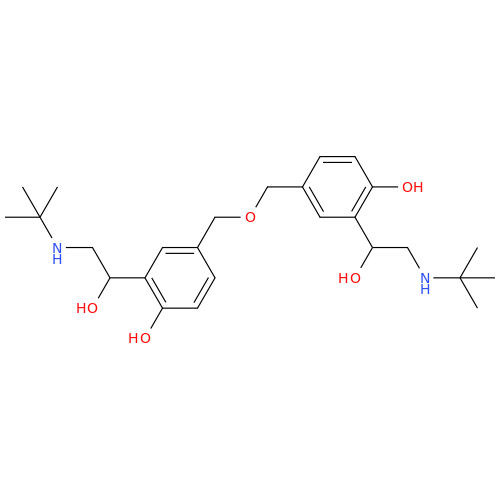
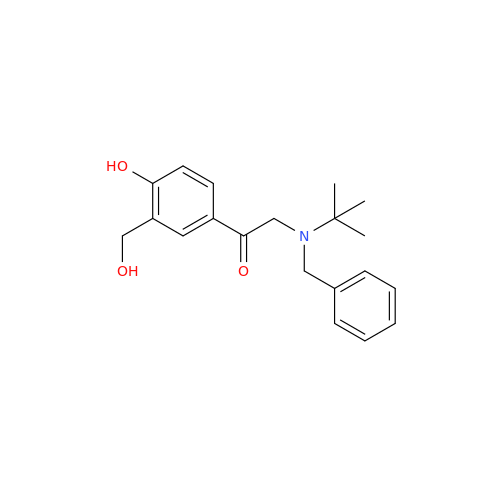
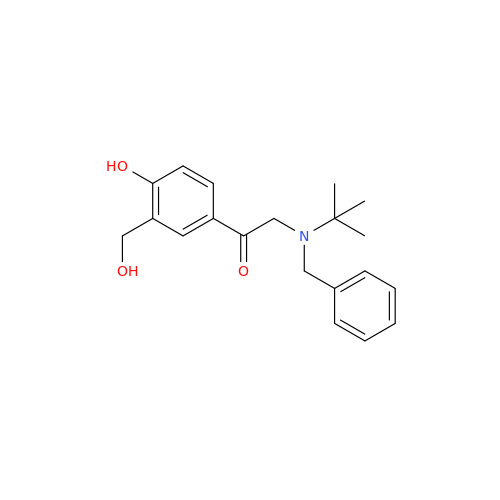
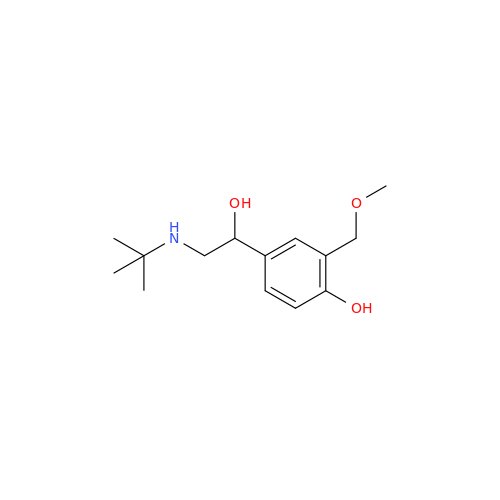
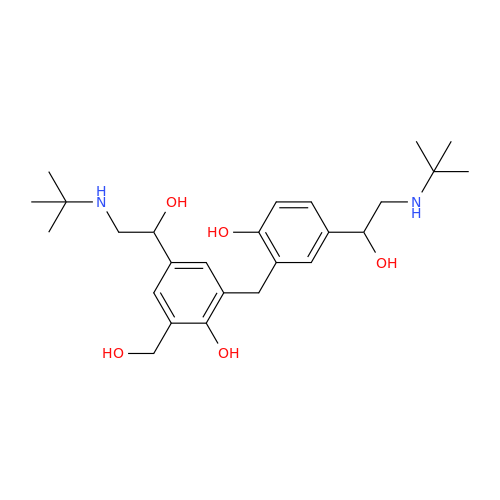
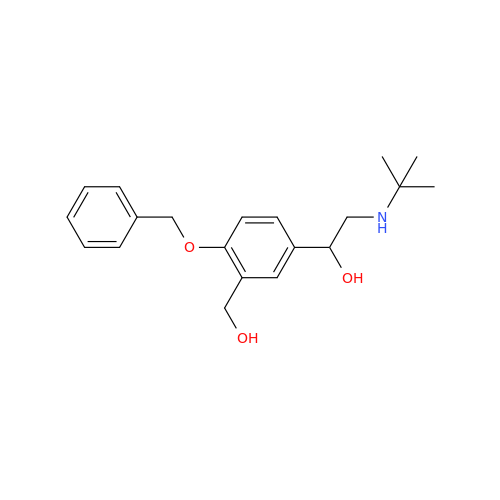
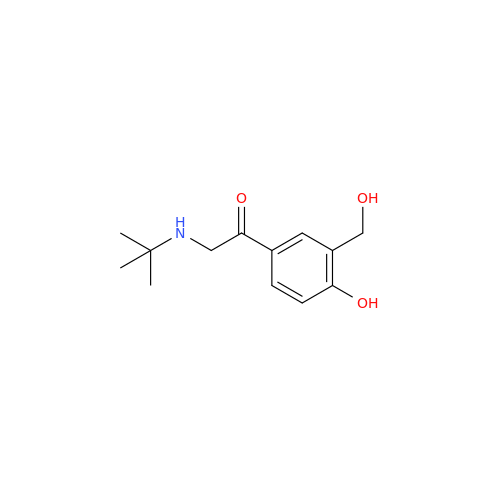
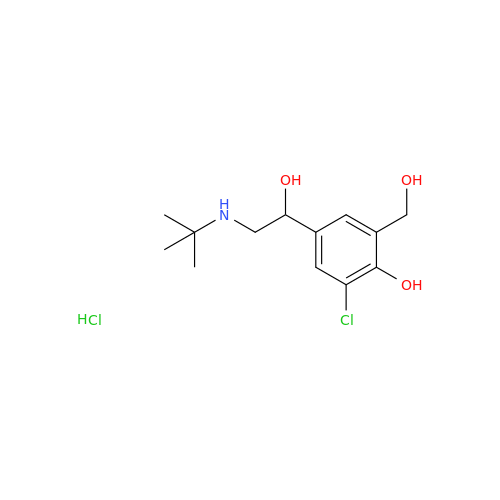
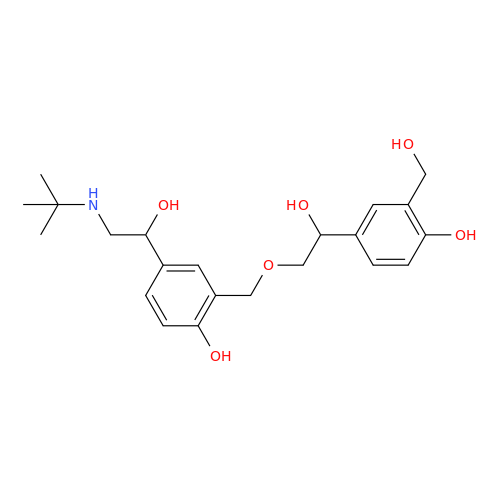
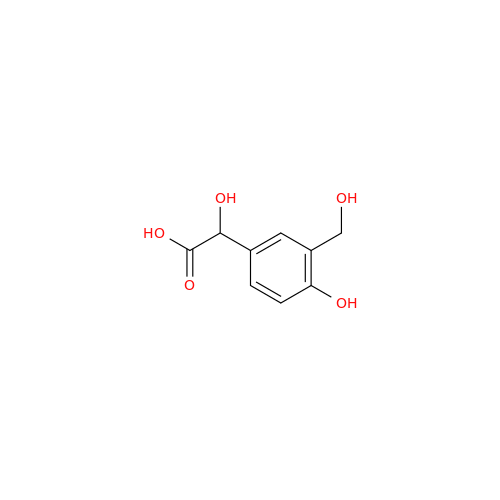
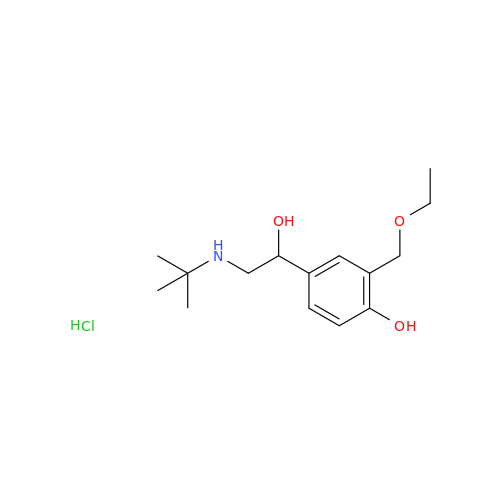
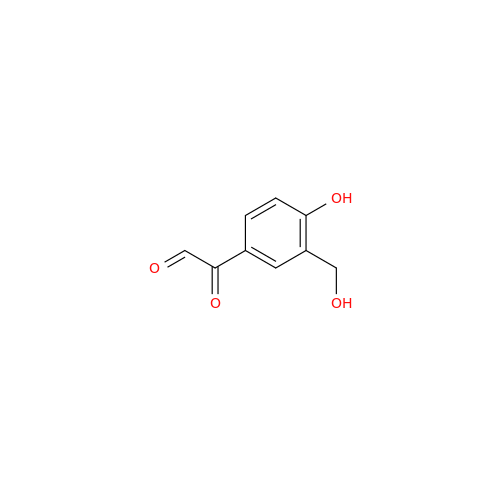
Salbutamol EP Impurity Q
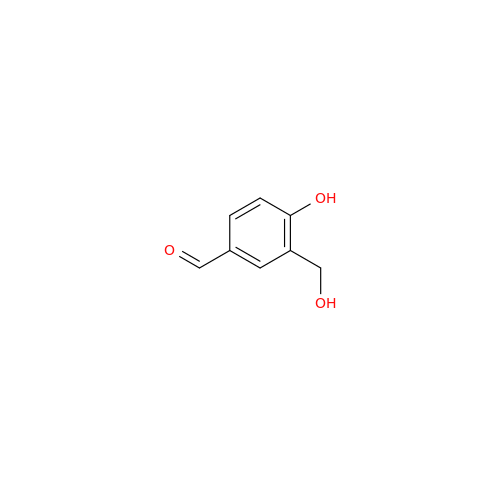
|
Chemical Name: Salbutamol EP Impurity Q
Synonym: 3-(Hydroxymethyl)-4-hydroxybenzaldehyde; Benzaldehyde, 4-hydroxy-3-(hydroxymethyl)-| Enter Batch Number | |||