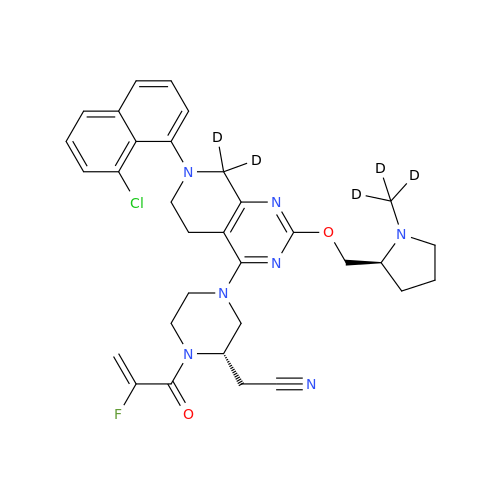
Product Information
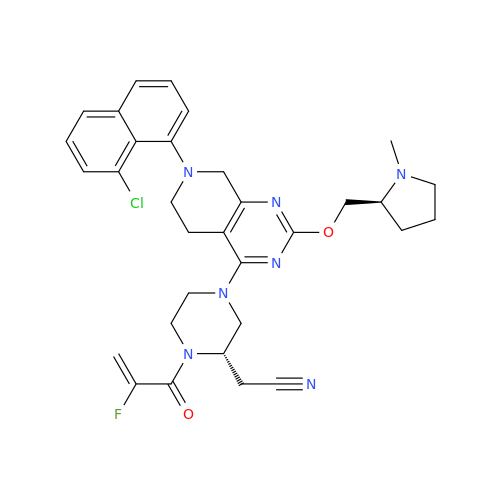
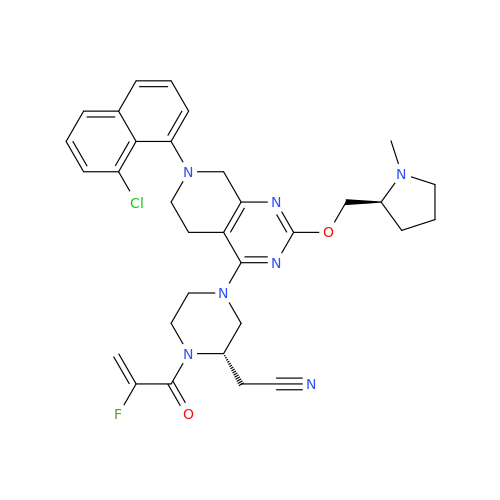
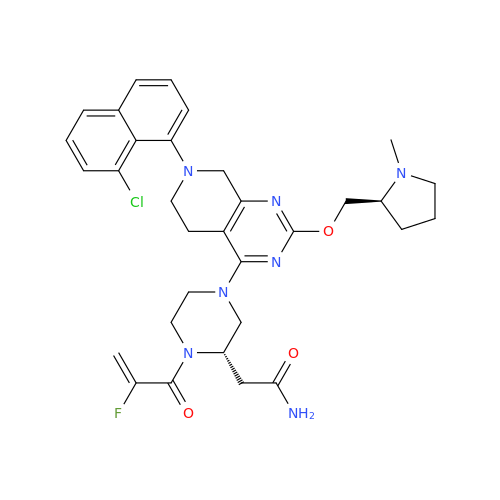
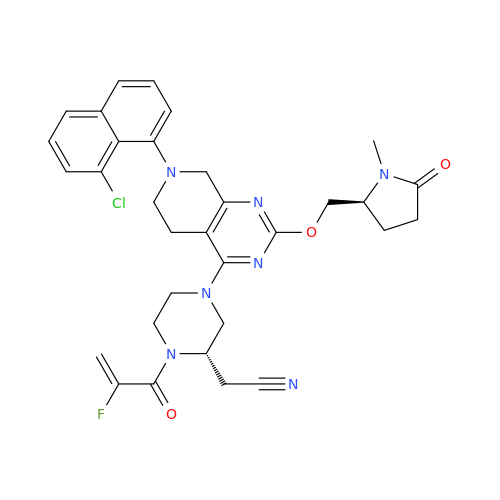
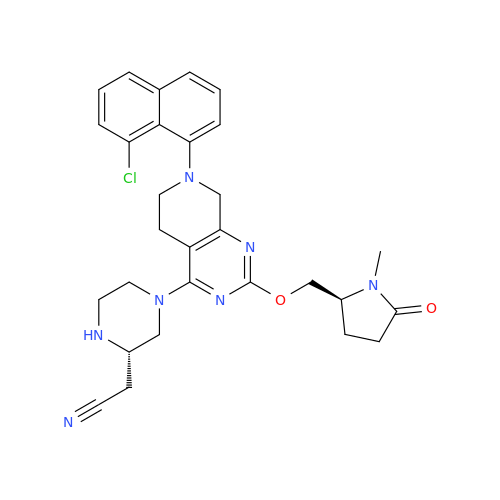
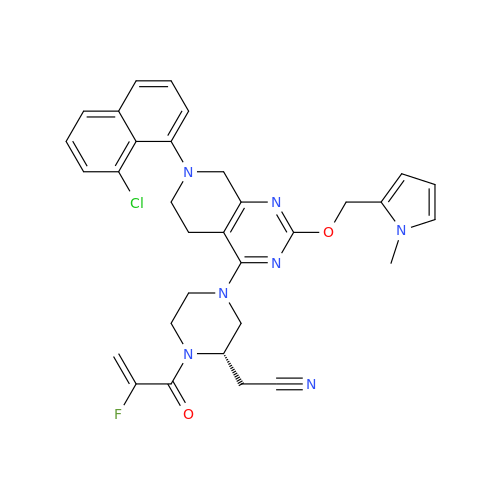
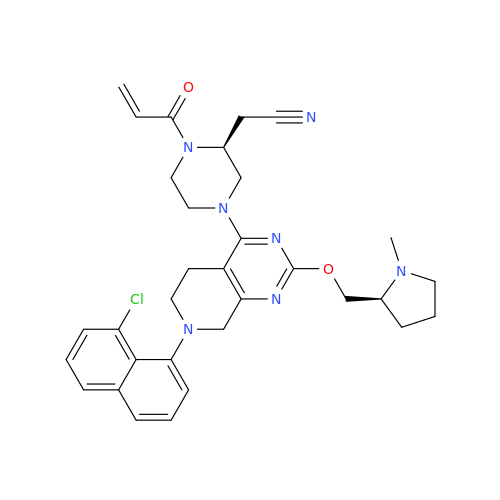
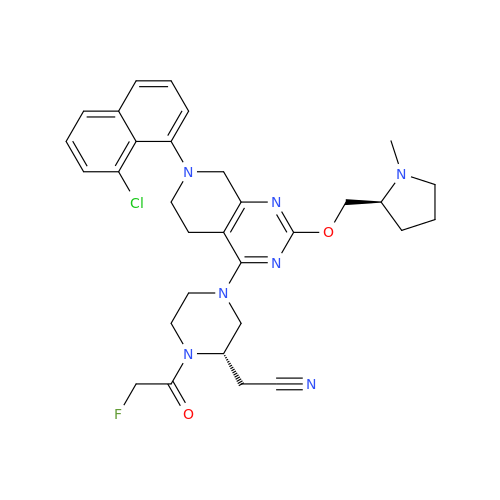
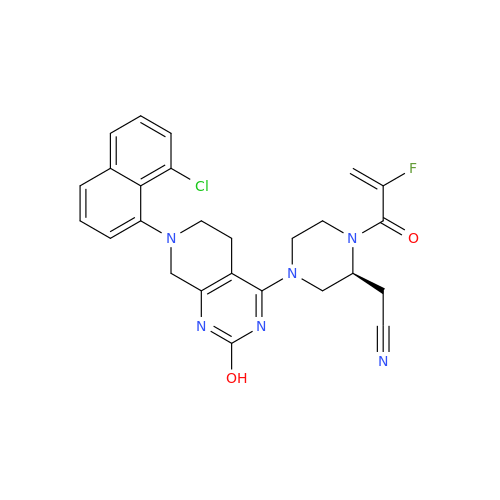
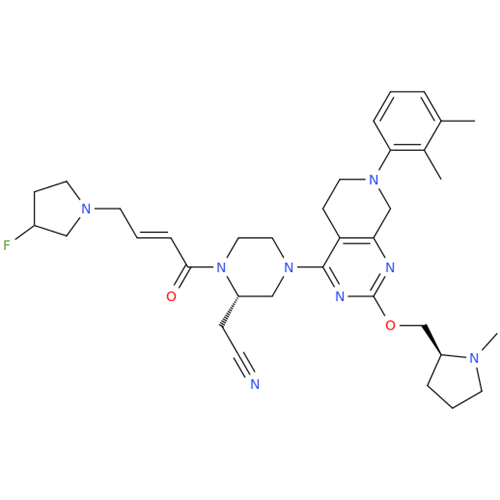
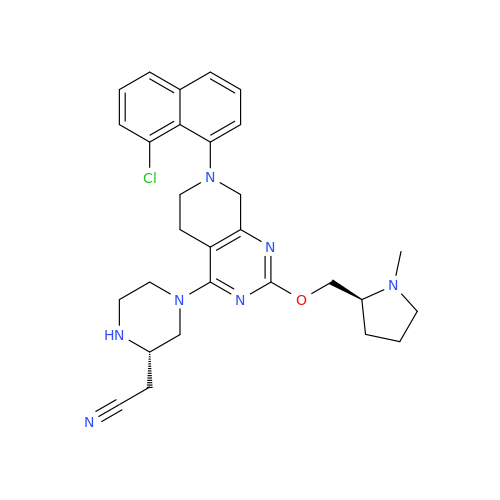
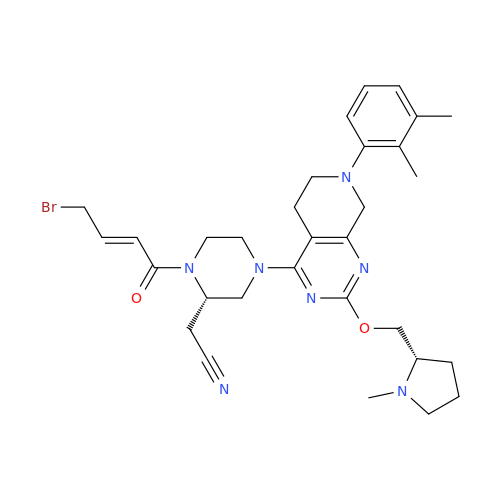
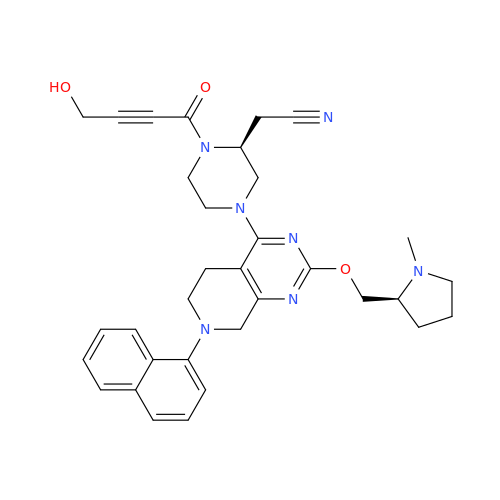
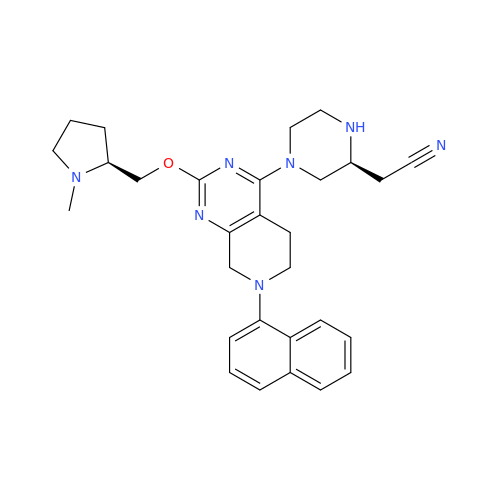
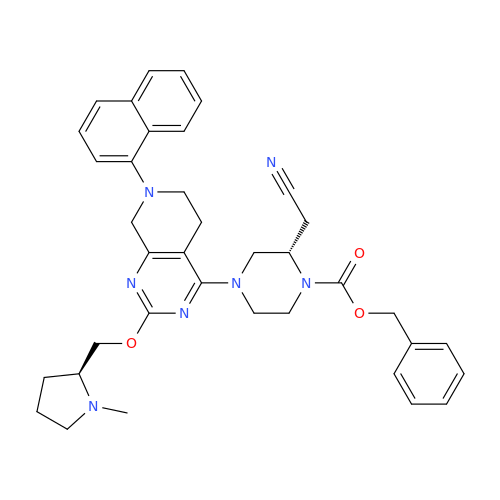
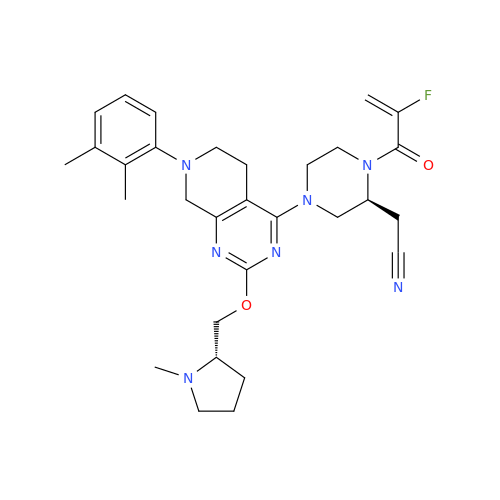
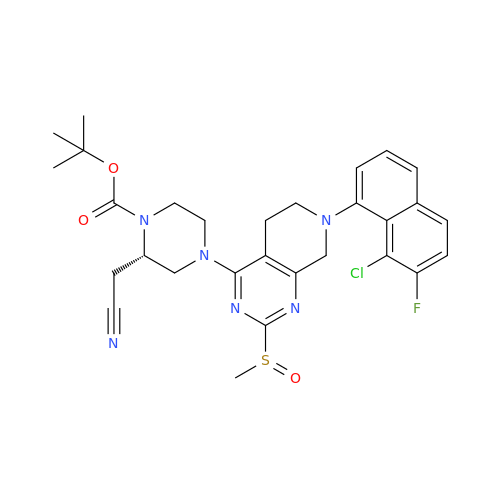
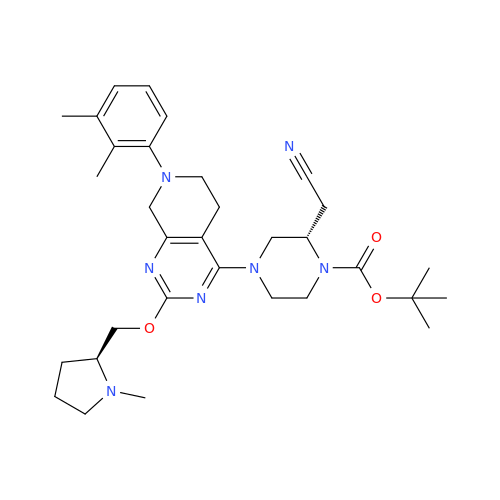
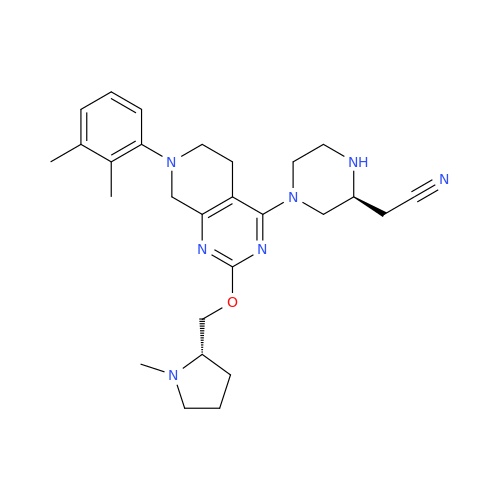
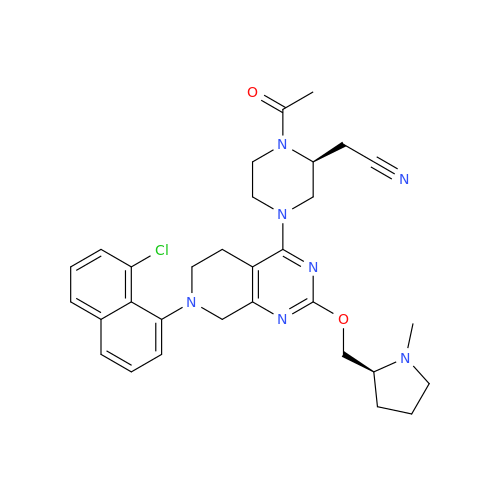
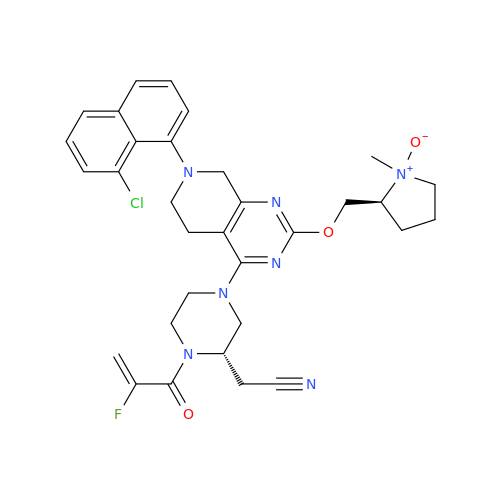
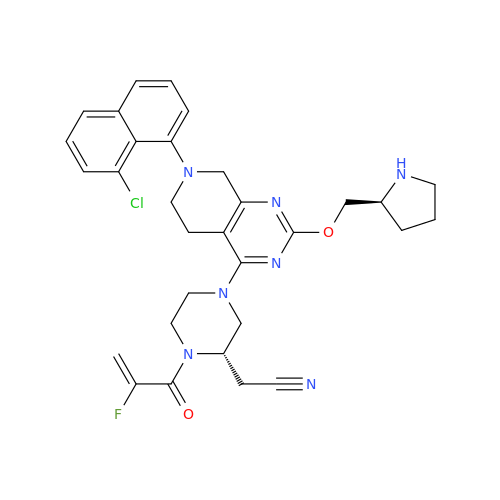
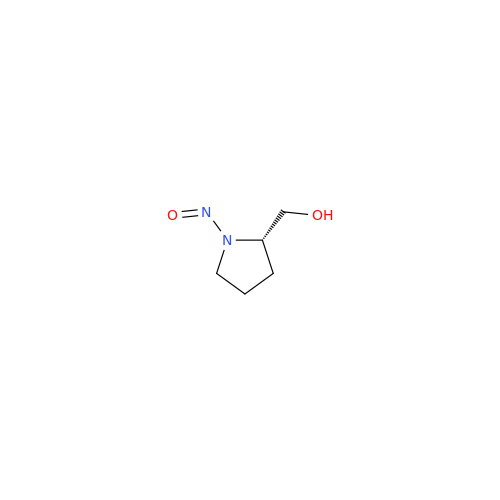
Adagrasib Impurity 11
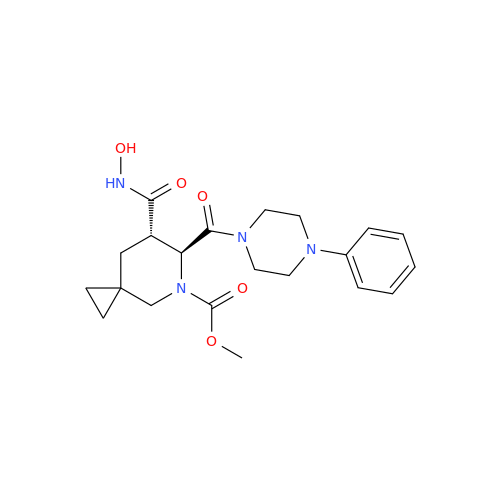
|
Chemical Name: Adagrasib Impurity 11
Synonym: methyl (6S,7S)-7-(hydroxycarbamoyl)-6-(4-phenylpiperazine-1-carbonyl)-5-azaspiro[2.5]octane-5-carboxylate| Enter Batch Number | |||