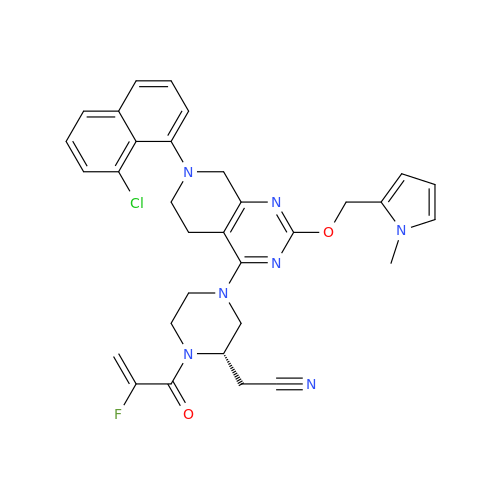
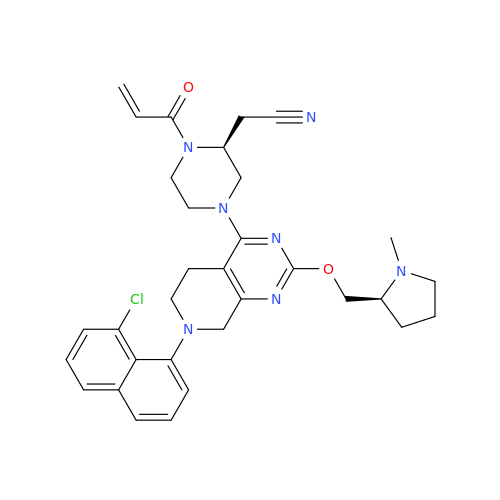
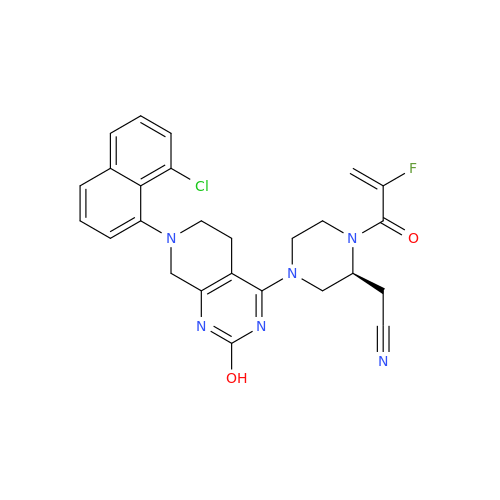
Product Information
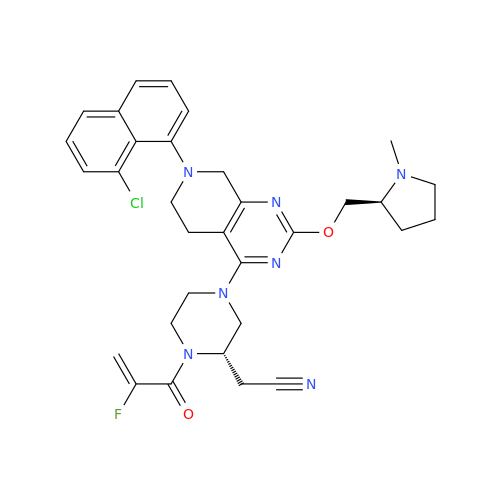
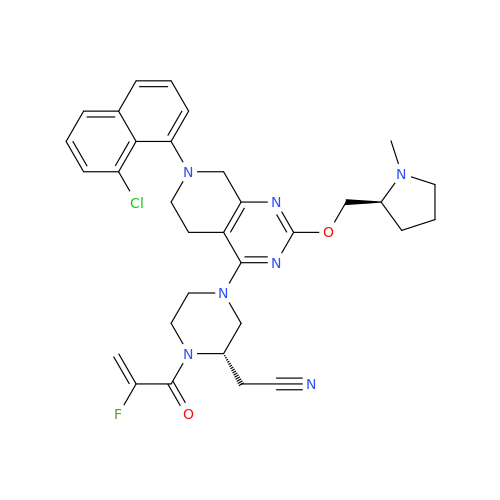
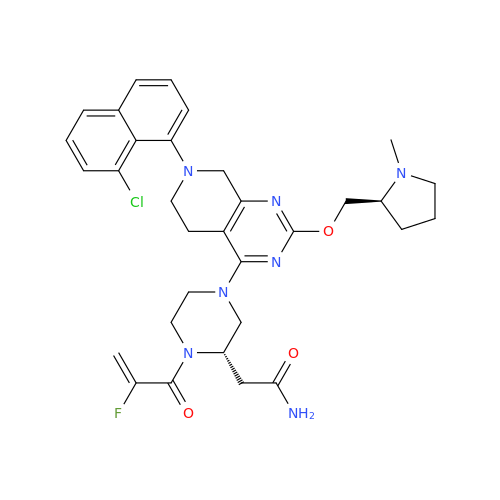
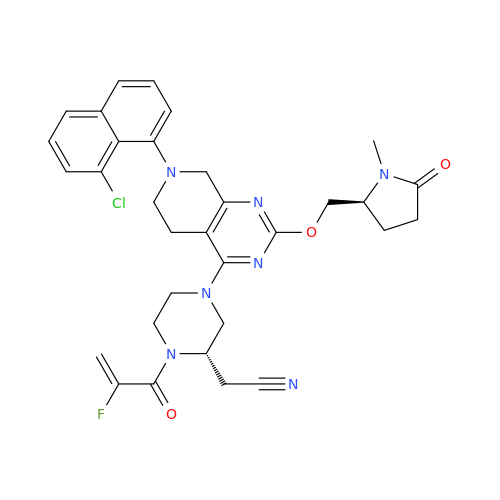
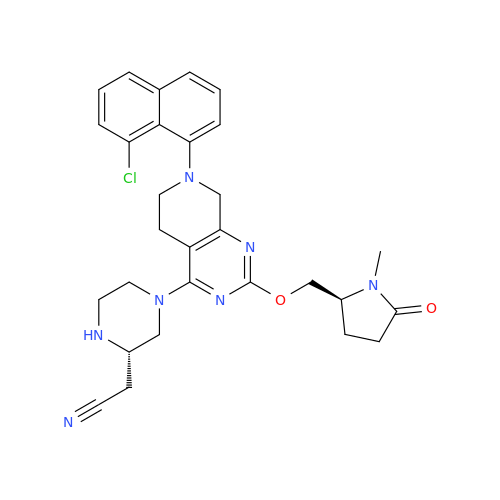
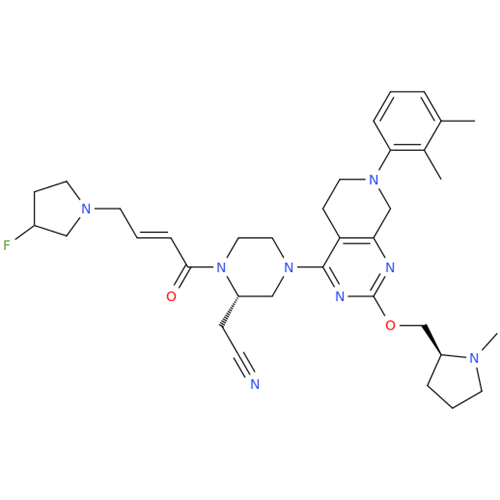
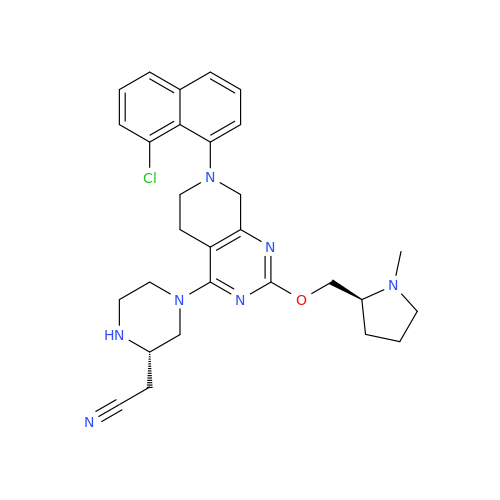
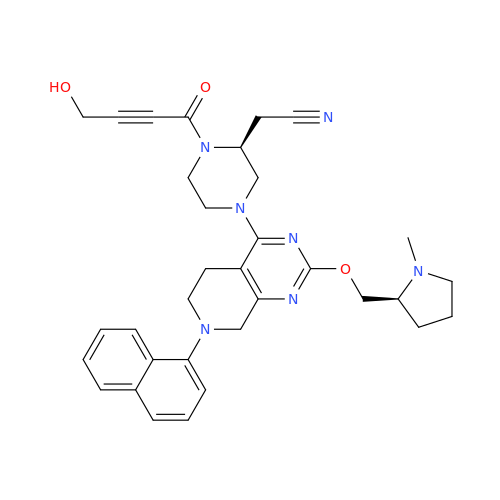
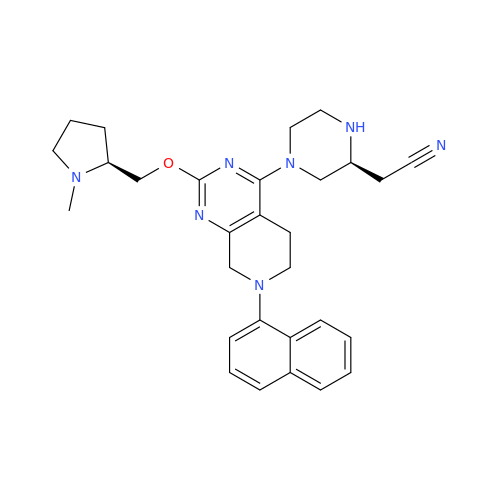
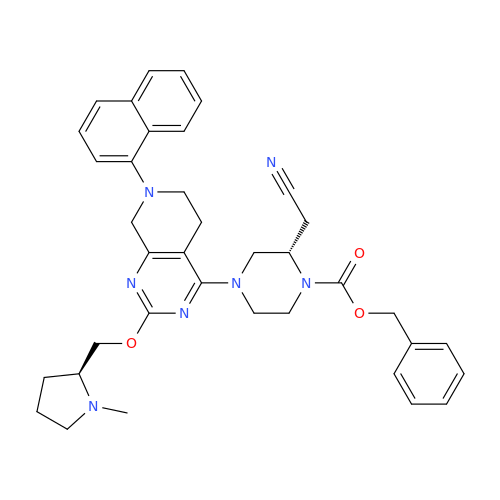
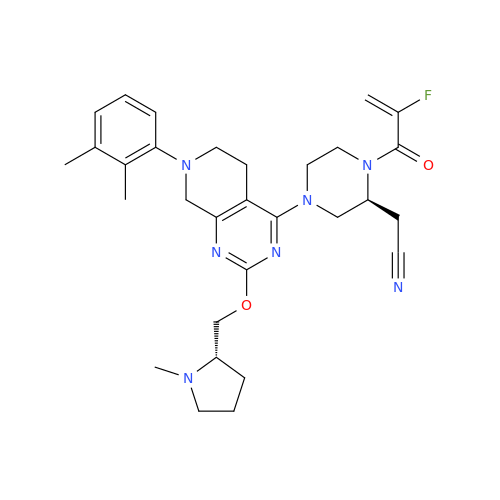
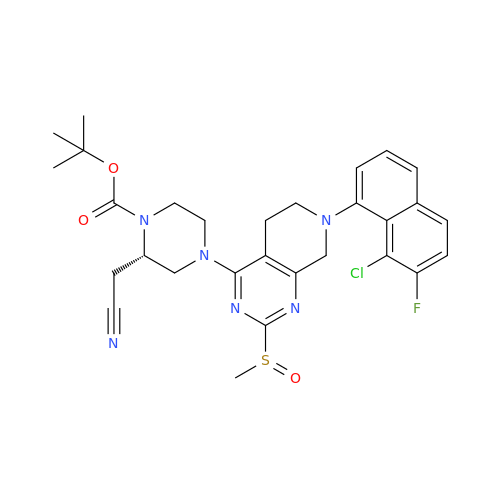
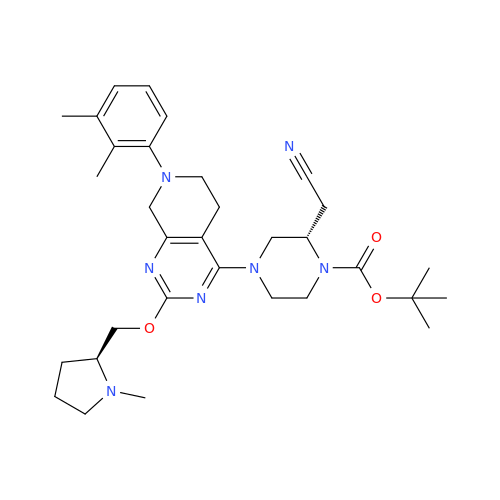
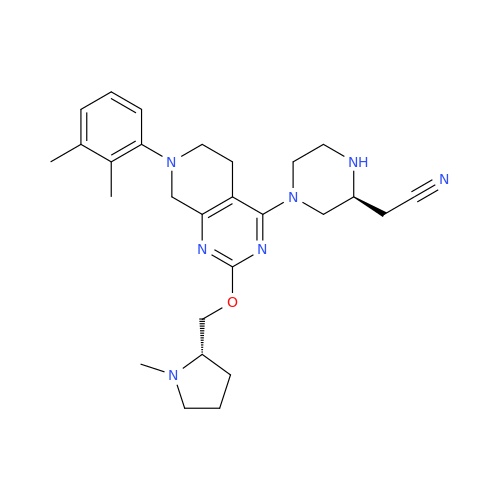
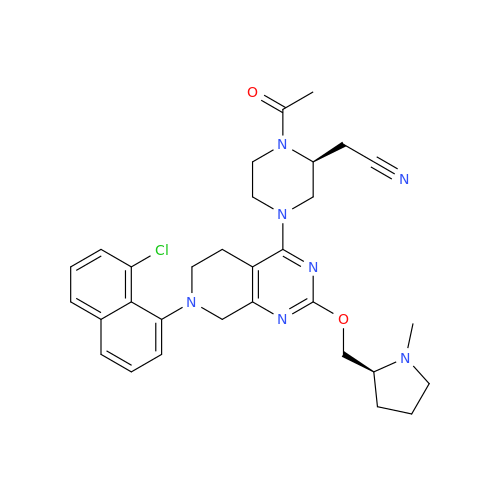
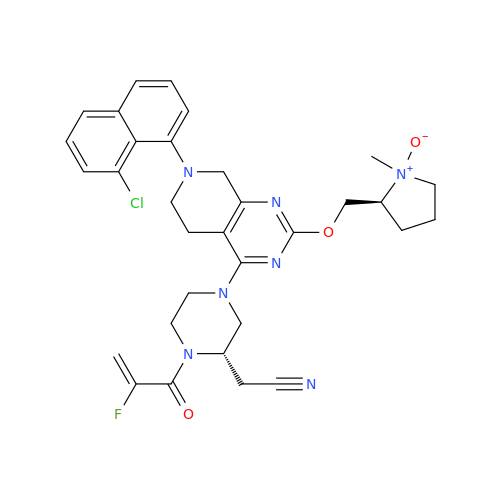
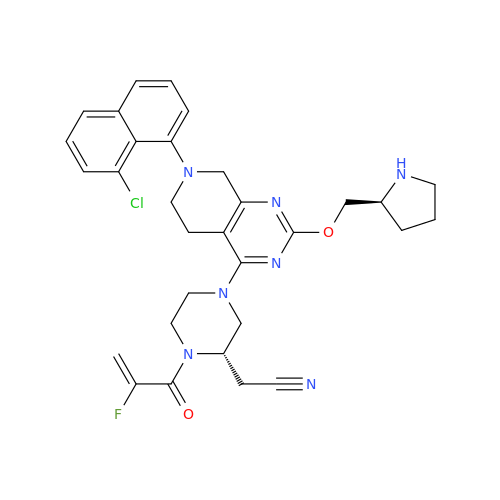
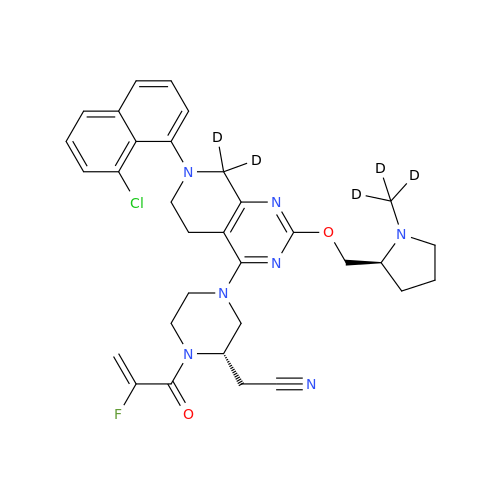
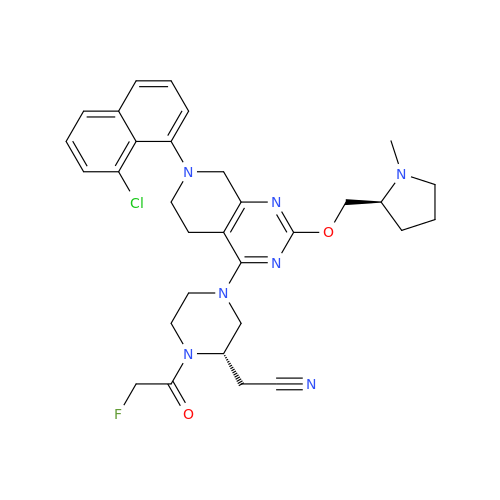
Adagrasib Desmethylene Impurity
|
Chemical Name: Adagrasib Desmethylene Impurity
Synonym: 2-((S)-4-(7-(8-Chloronaphthalen-1-yl)-2-(((S)-1-methylpyrrolidin-2-yl)methoxy)-5,6,7,8-tetrahydropyrido[3,4-d]pyrimidin-4-yl)-1-(2-fluoroacetyl)piperazin-2-yl)acetonitrile| Enter Batch Number | |||