Product Information
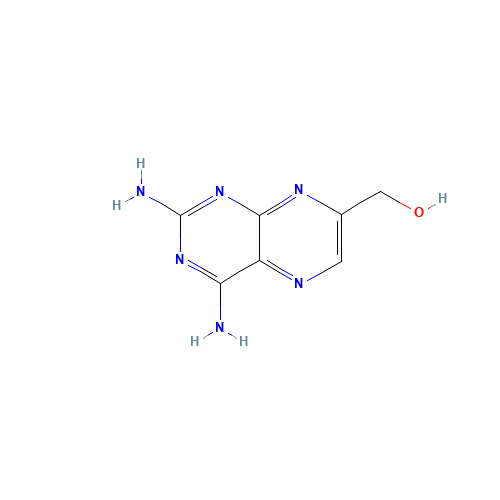
Methotrexate Impurity 12
|
Chemical Name: Methotrexate Impurity 12
Synonym:| Enter Batch Number | |||

|
Chemical Name: Methotrexate Impurity 12
Synonym:| Enter Batch Number | |||