Product Information
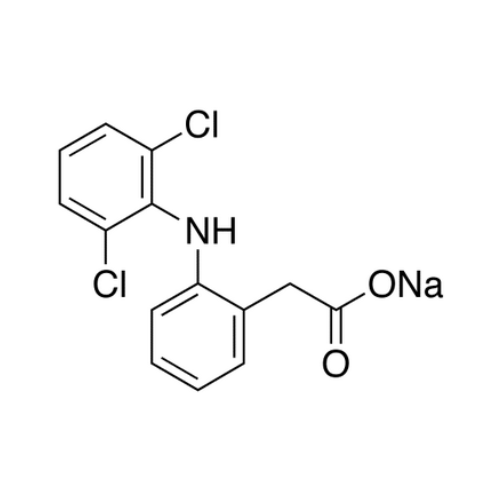
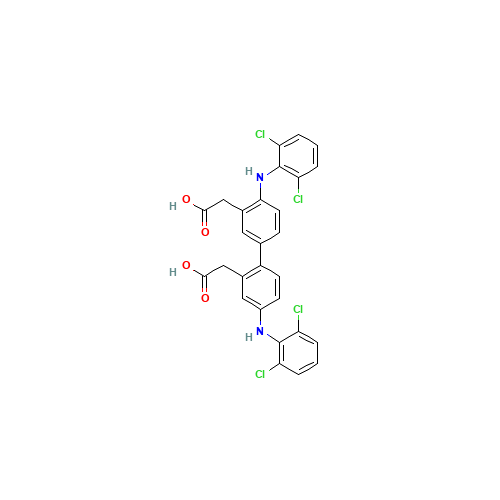
Diclofenac EP Impurity B
|
Chemical Name: Diclofenac EP Impurity B
Synonym: Diclofenac Benzaldehyde Analog| Enter Batch Number | |||