Product Information
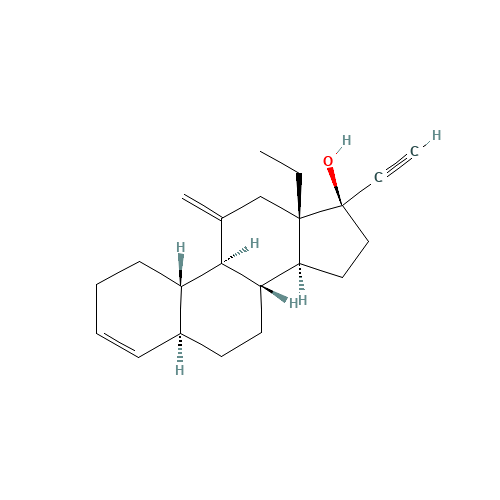
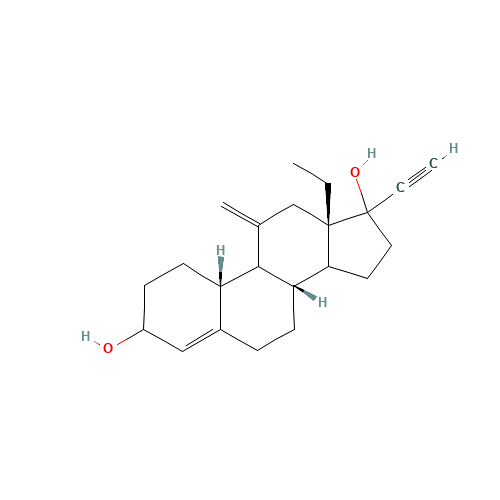
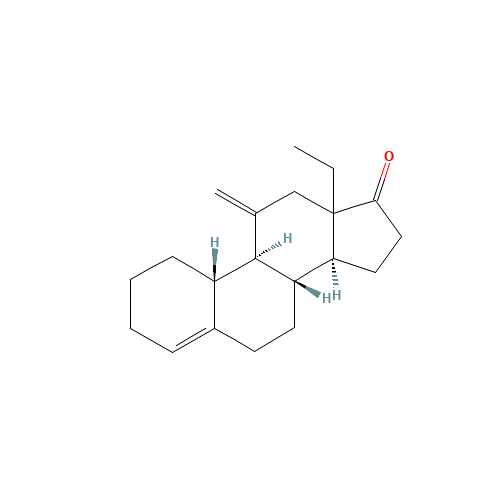
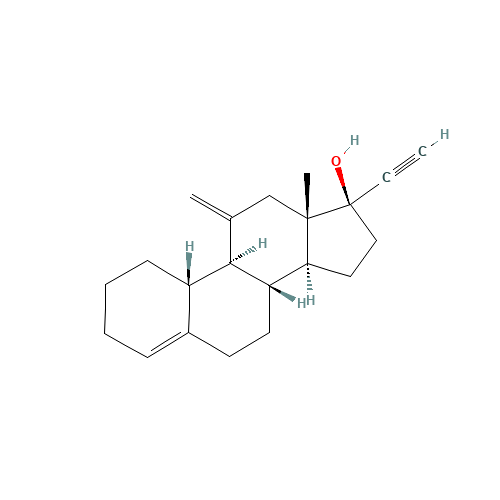
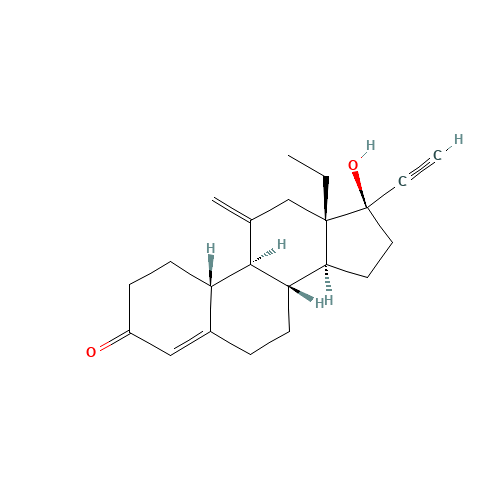
Desogestrel EP Impurity D
|
Chemical Name: Desogestrel EP Impurity D
Synonym: Desogestrel USP Related Compound C ; 3-Ketodesogestrel ; Etonogestrel| Enter Batch Number | |||