Product Information
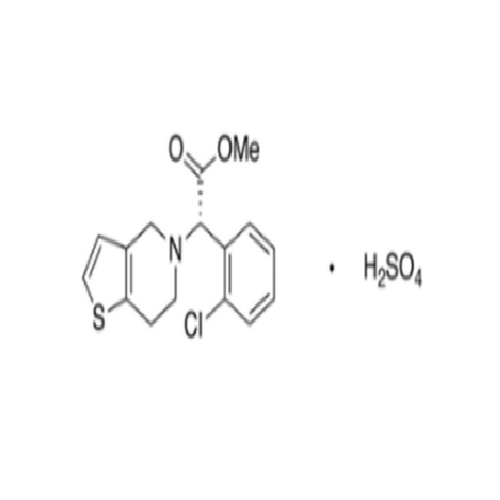
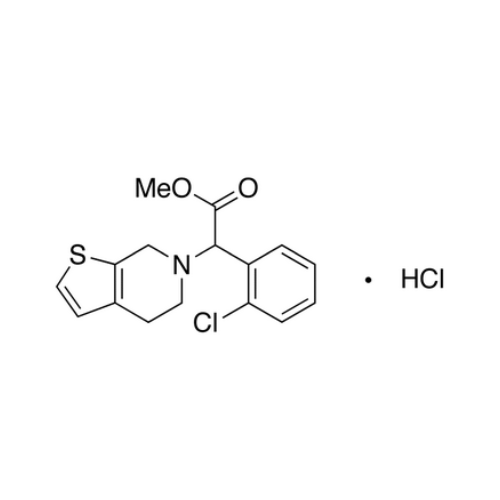
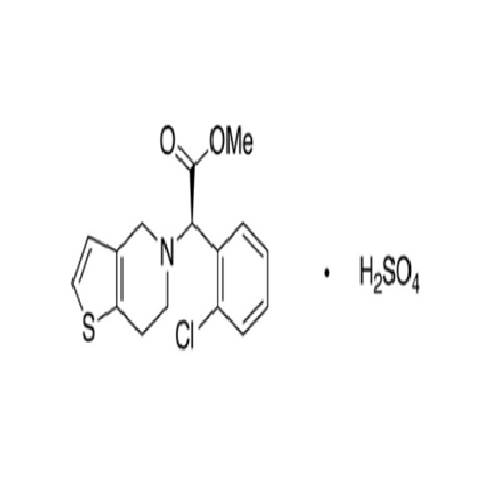
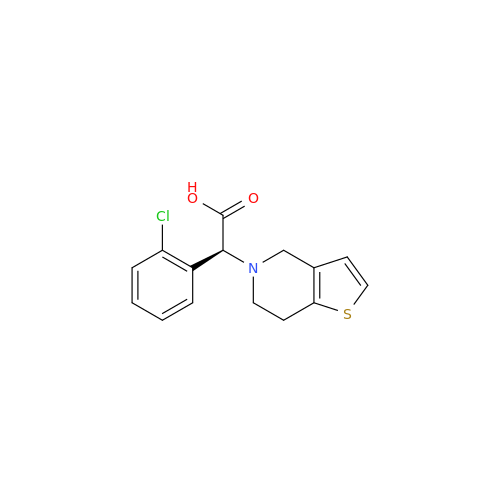
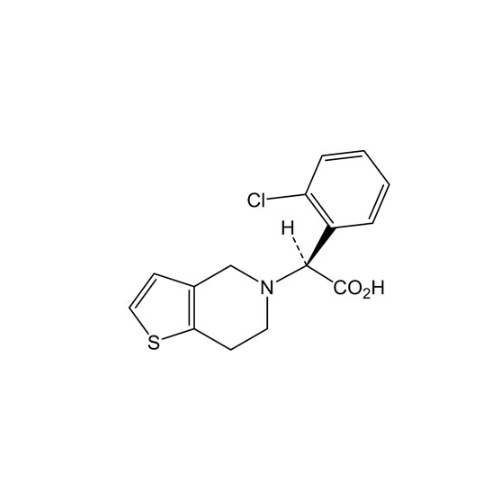
Clopidogrel EP Impurity A
|
Chemical Name: Clopidogrel EP Impurity A
Synonym: Clopidogrel USP Related Compound A; (S)-α-(2-Chlorophenyl)-6,7-dihydrothieno[3, 2-c]pyridine-5(4H)-acetic acid; SR 26334;| Enter Batch Number | |||