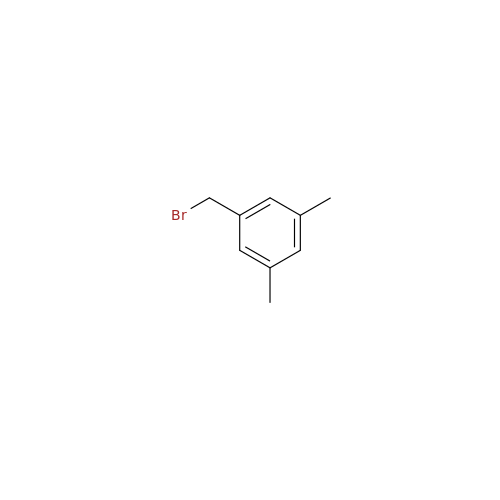
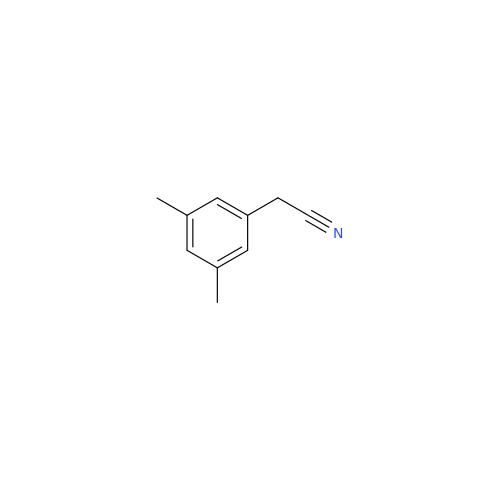
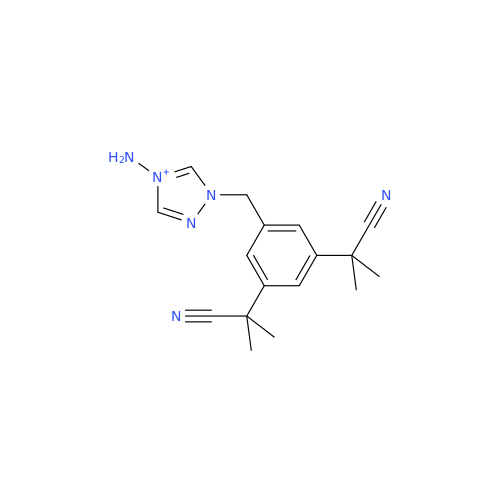
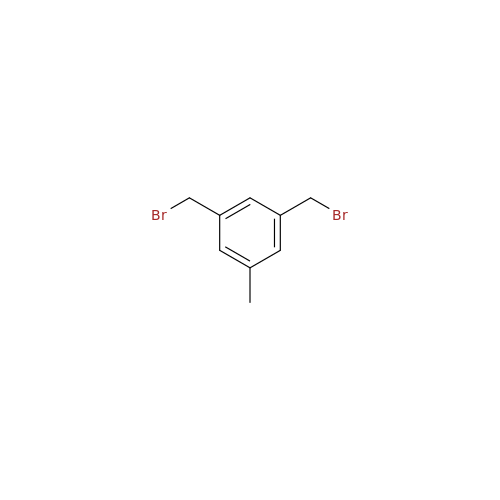
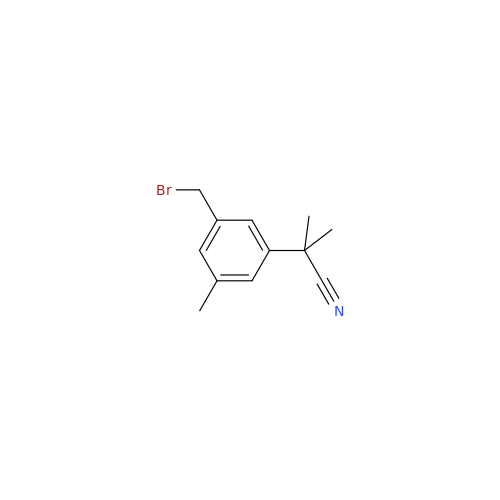
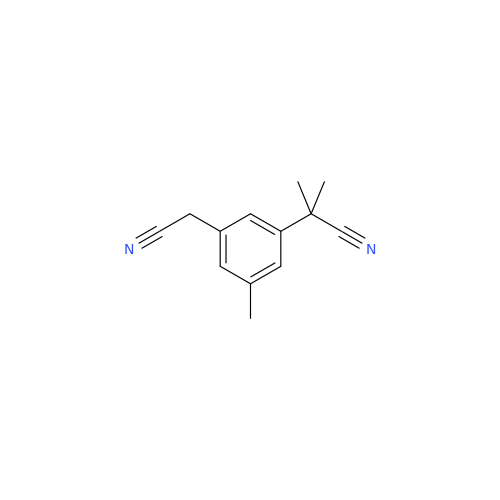
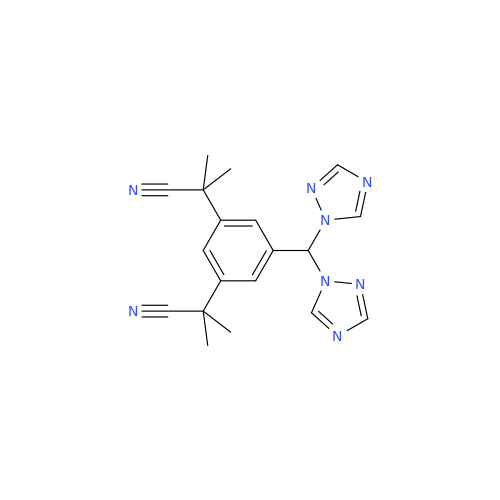
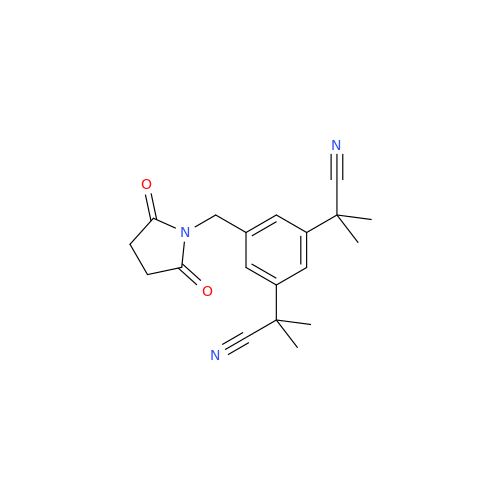
Product Information
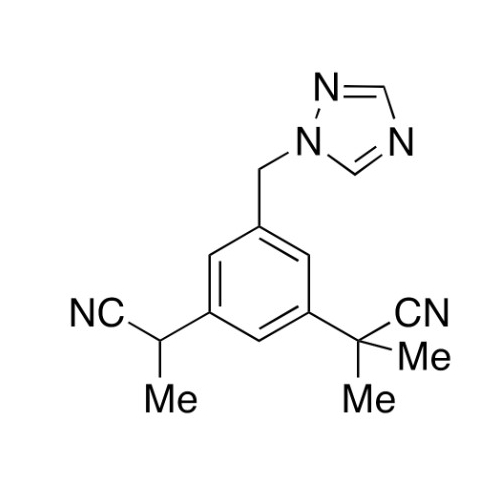
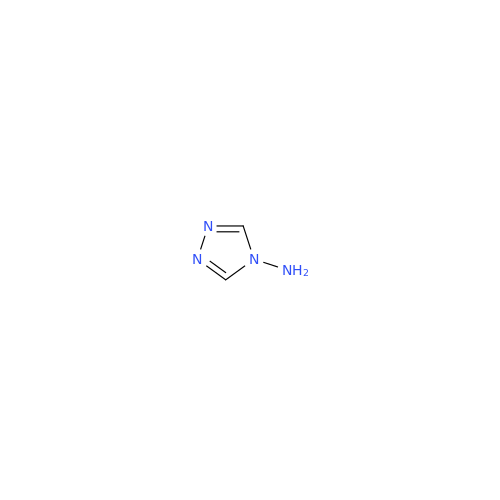
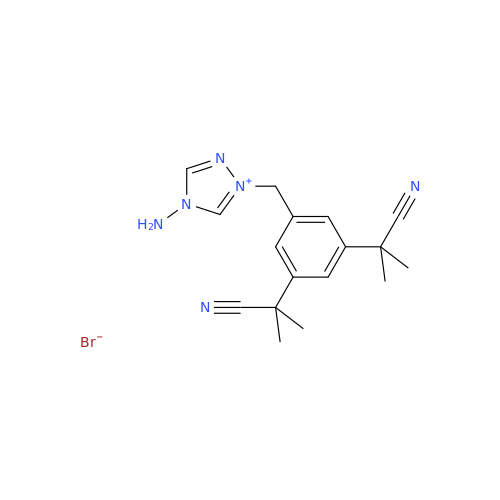
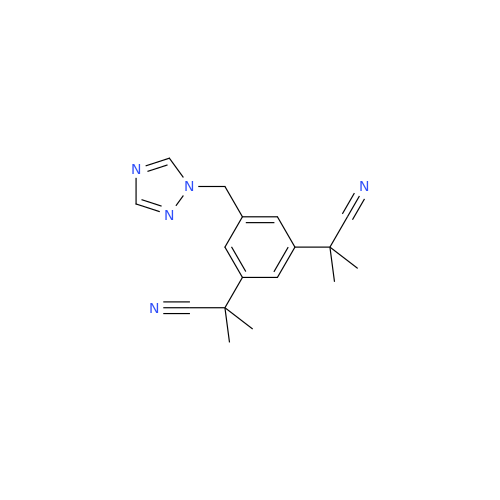
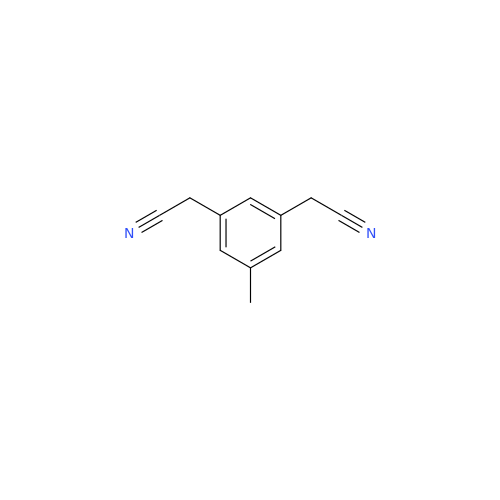
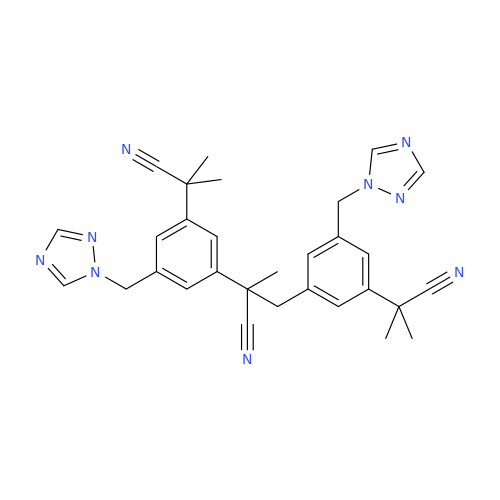
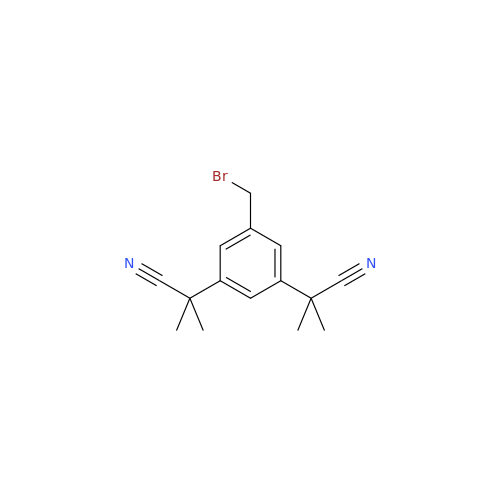
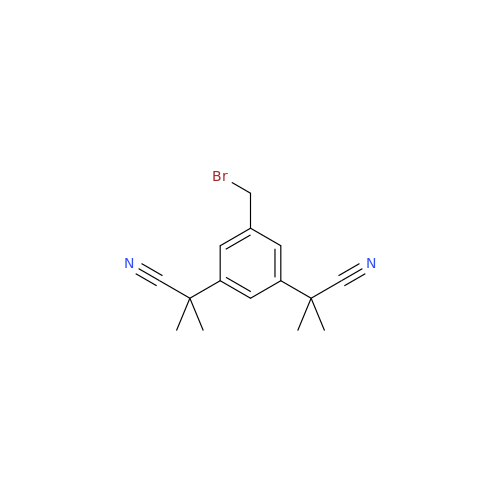
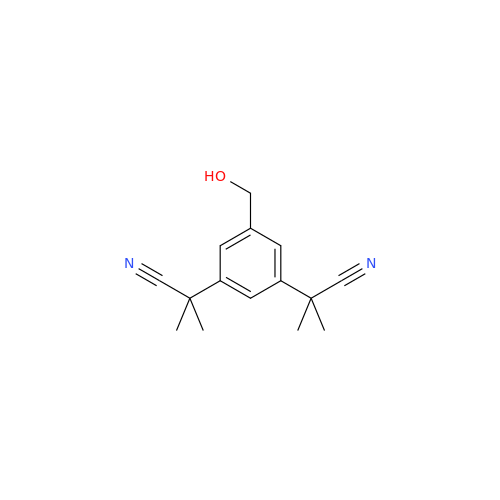
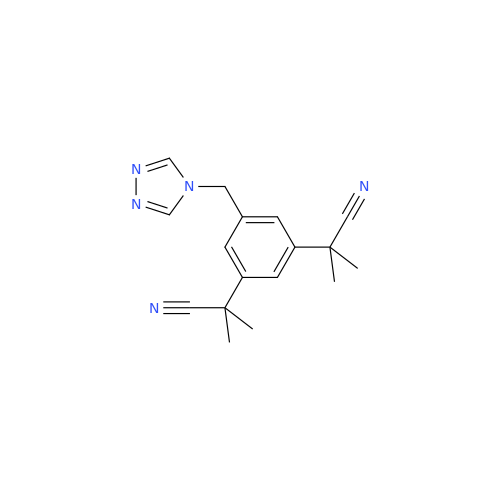
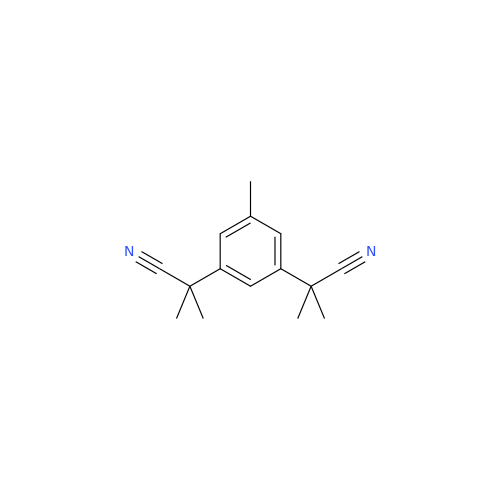
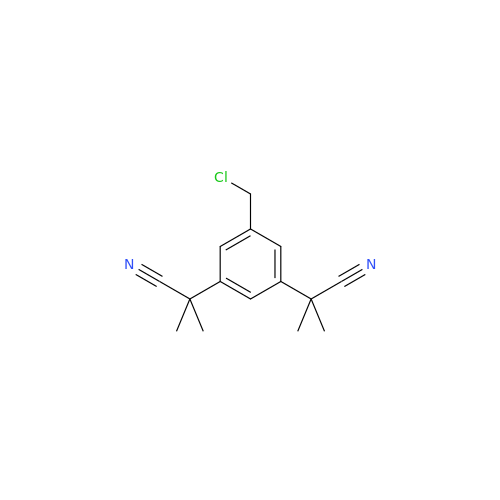
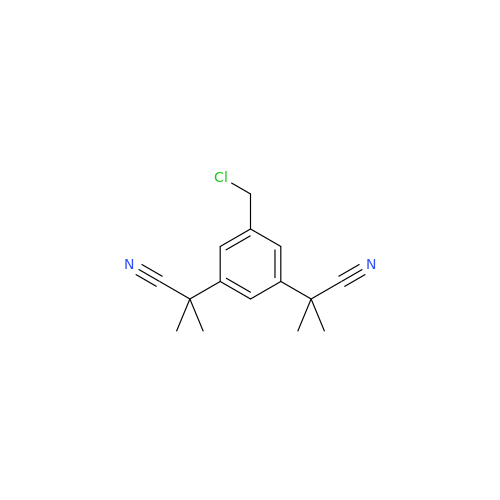
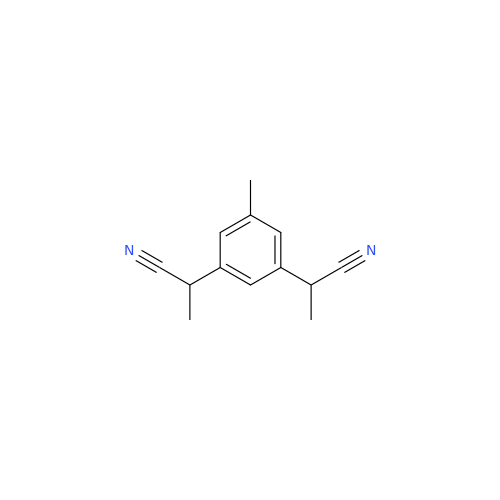
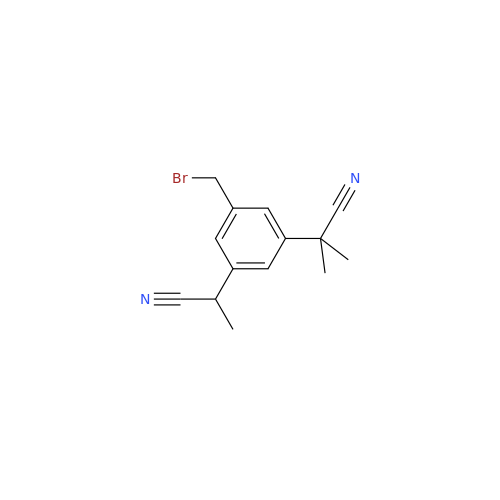
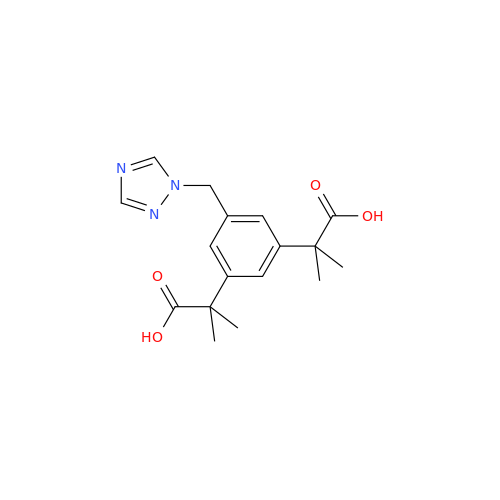
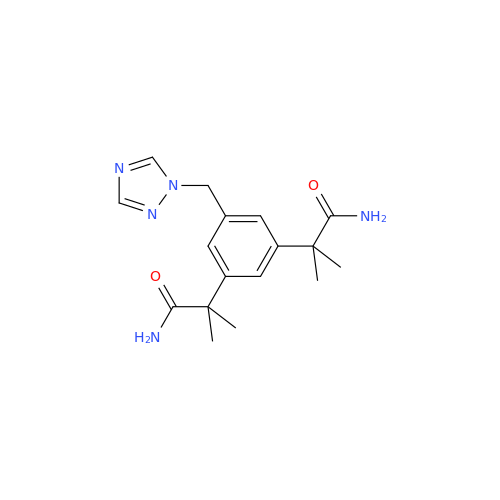
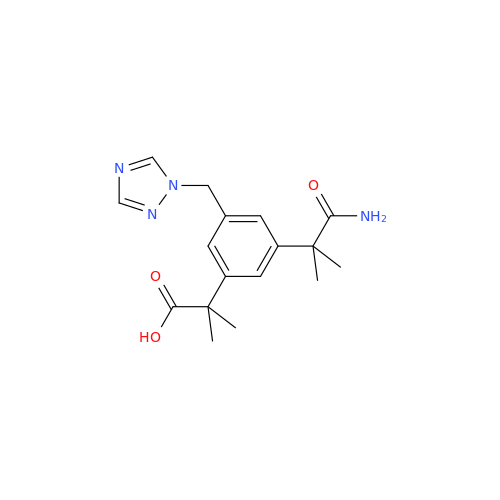
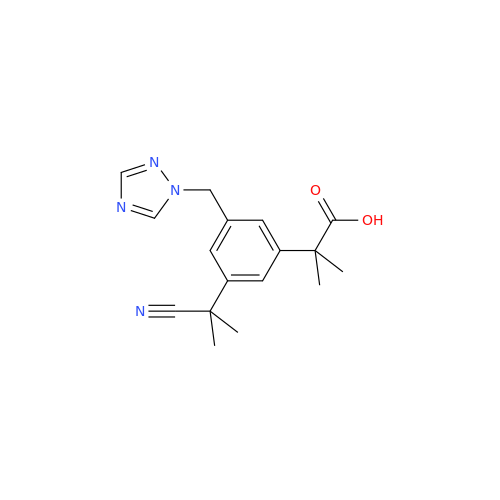
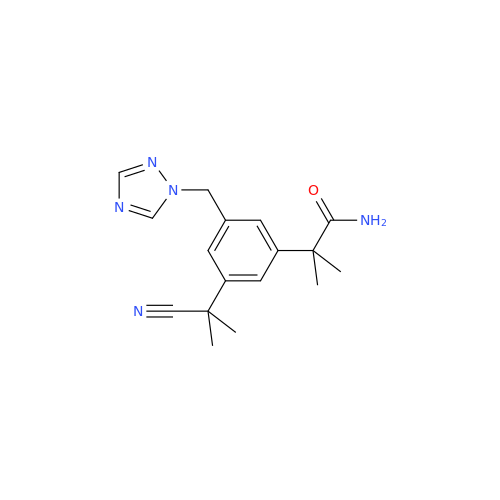
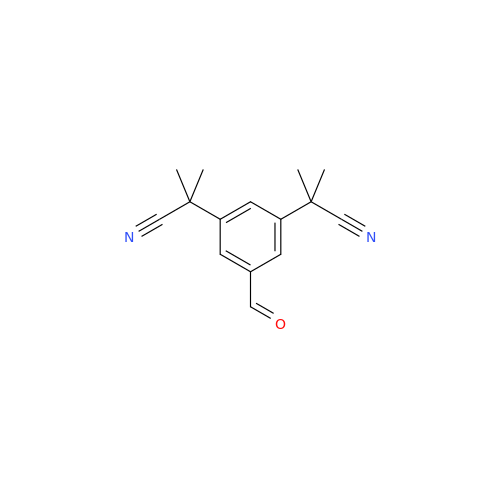
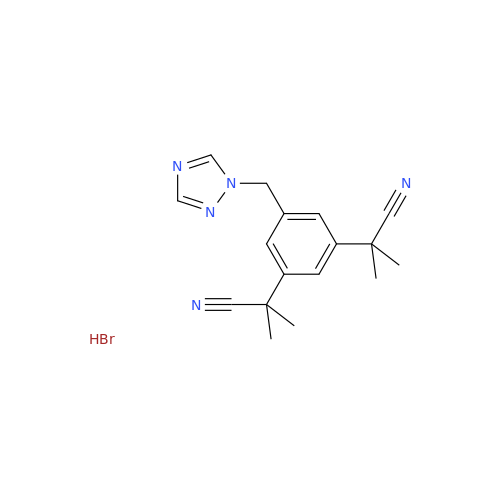
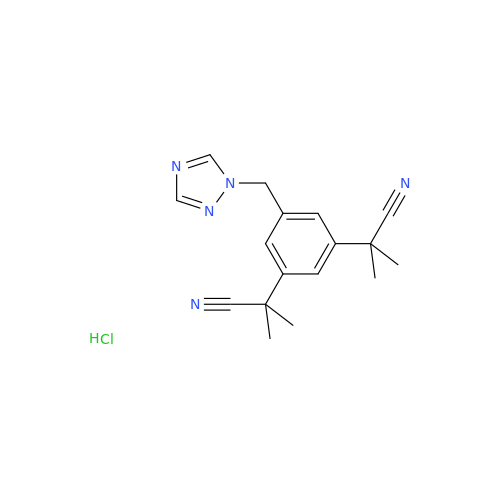
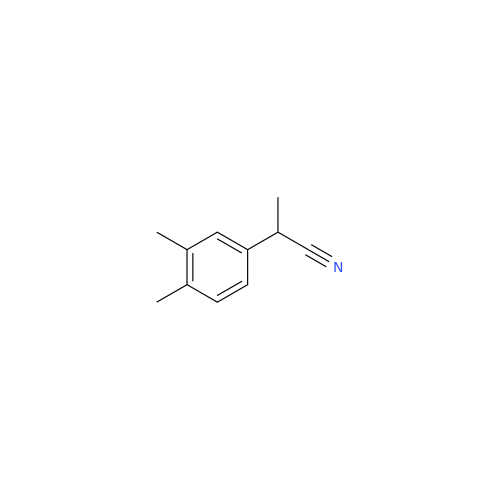
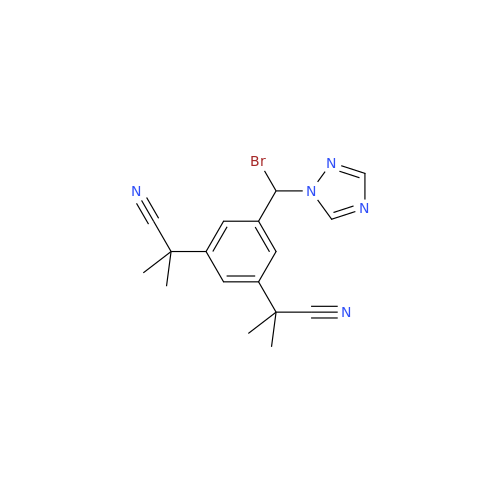
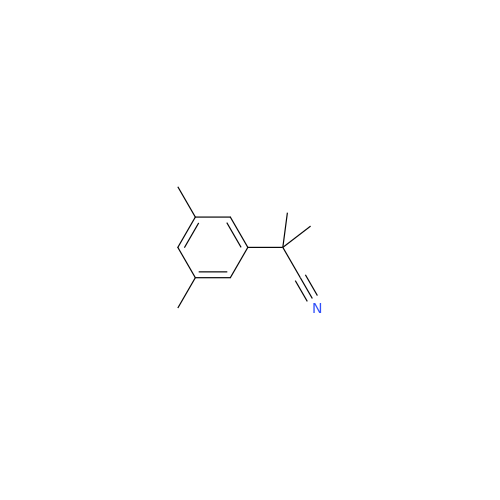
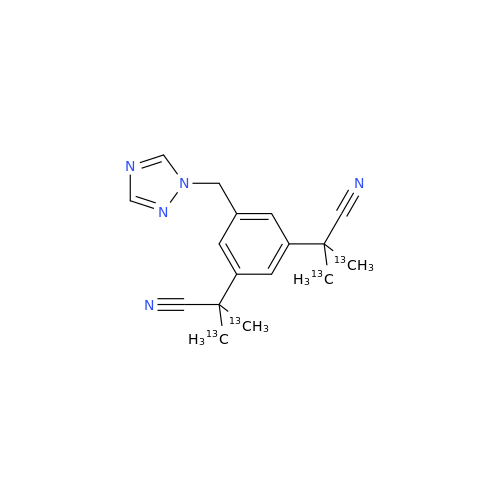
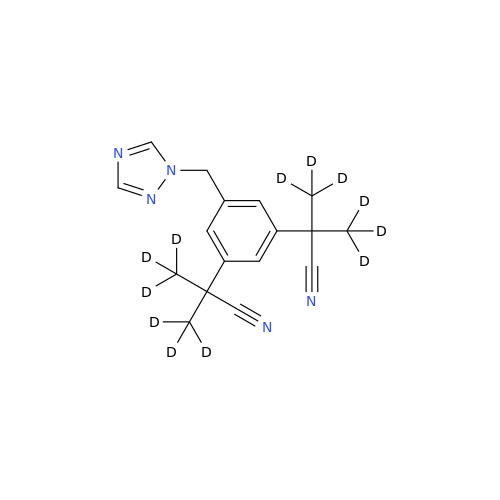
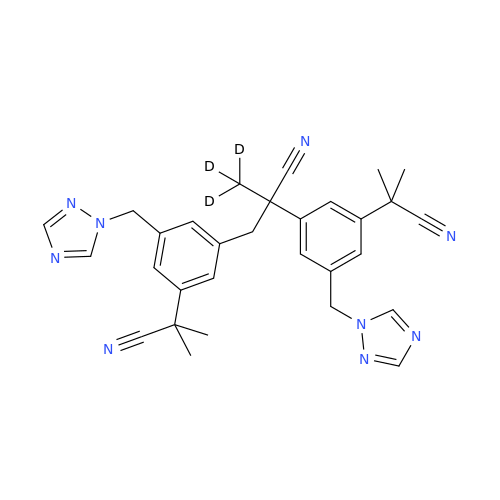
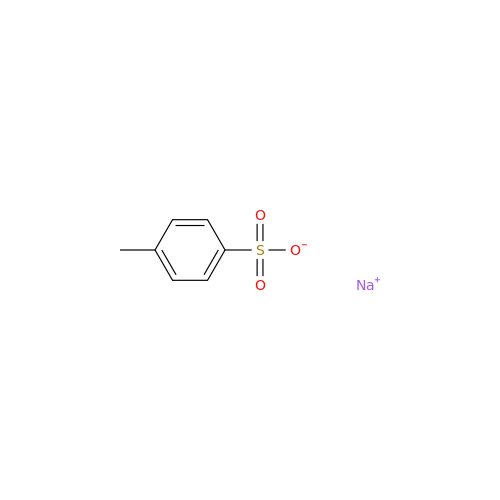
Anastrozole EP Impurity F (Sodium salt)
|
Chemical Name: Anastrozole EP Impurity F (Sodium salt)
Synonym: sodium 4-methylbenzenesulfonate| Enter Batch Number | |||