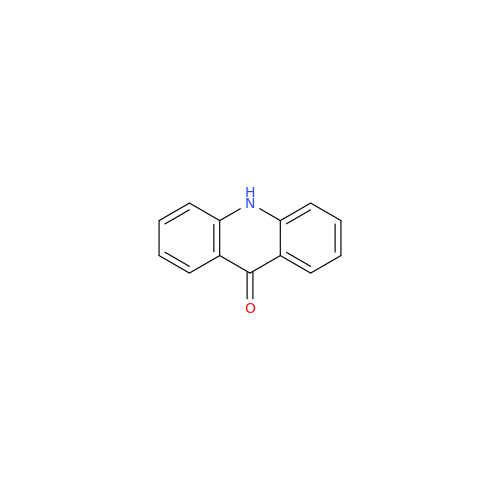
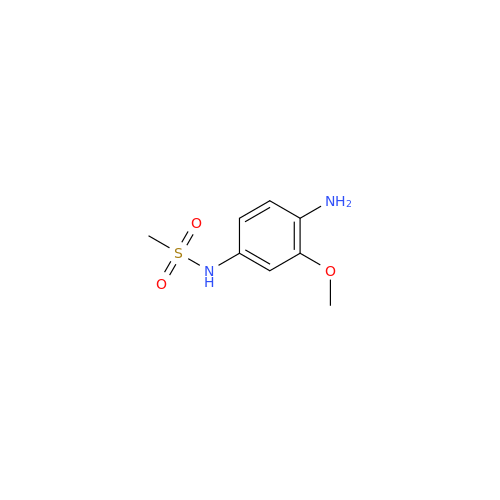
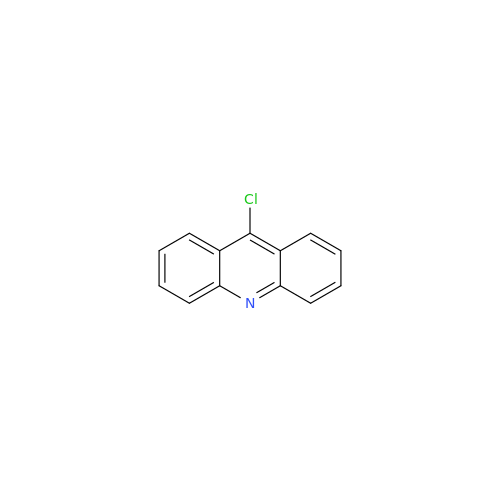
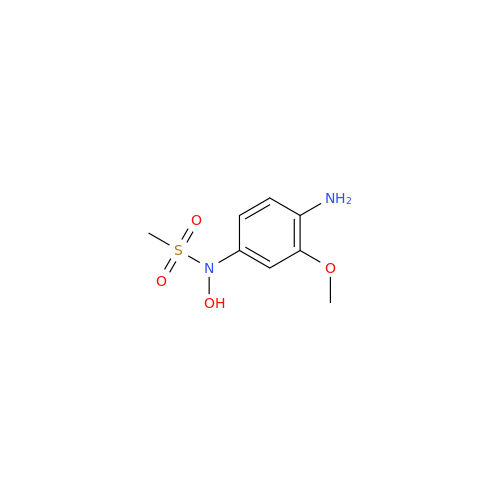
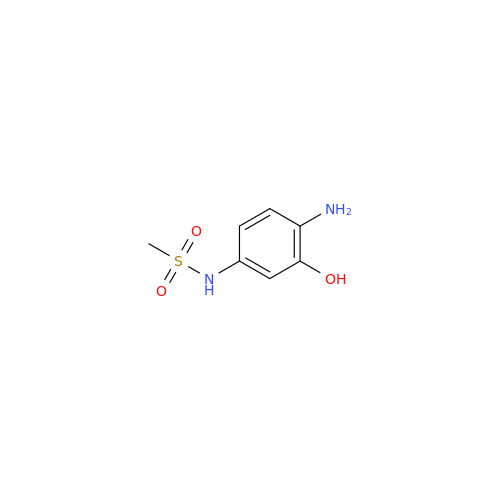
Product Information
Amsacrine IHRS
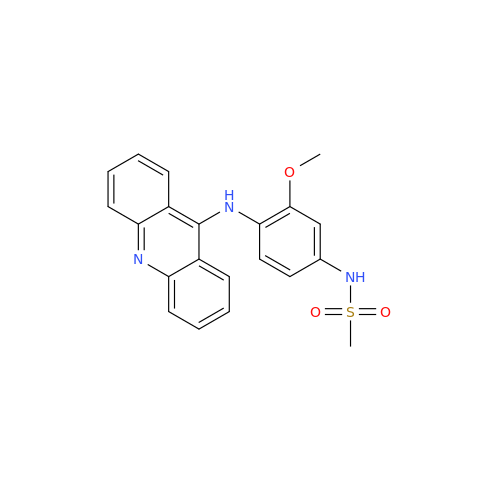
|
Chemical Name: Amsacrine IHRS
Synonym: N-(4-(Acridin-9-ylamino)-3-methoxyphenyl)methanesulfonamide| Enter Batch Number | |||