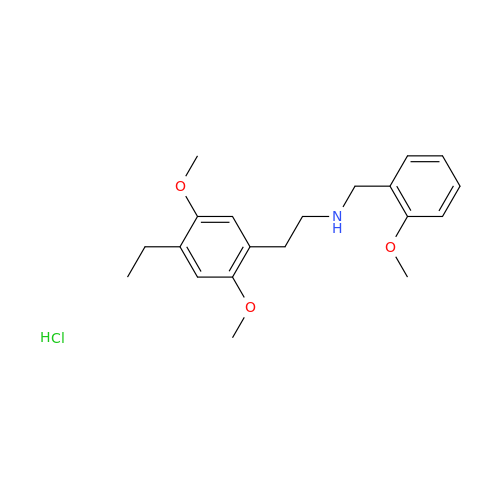
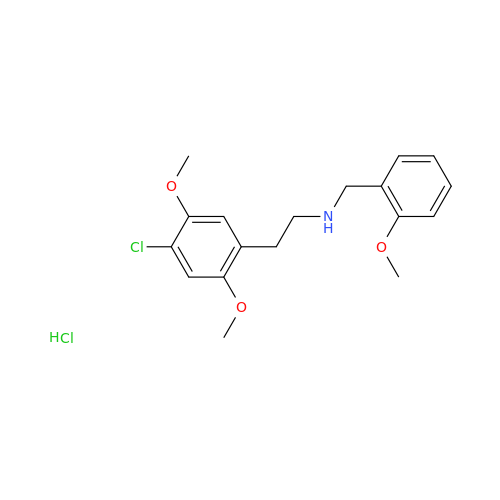
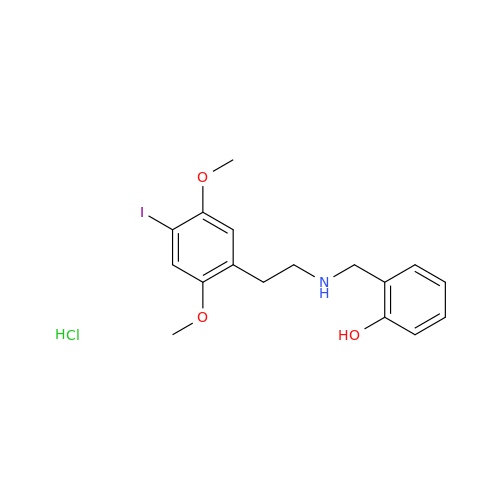
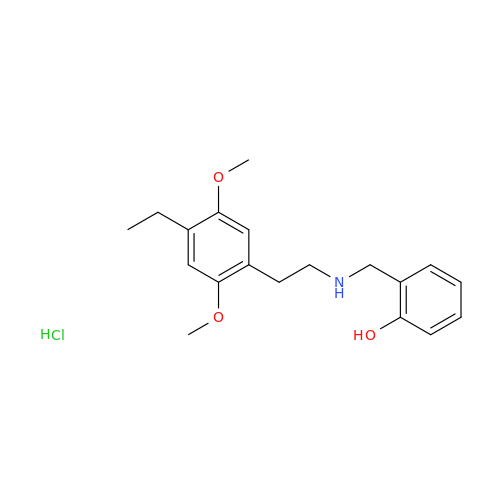
Product Information
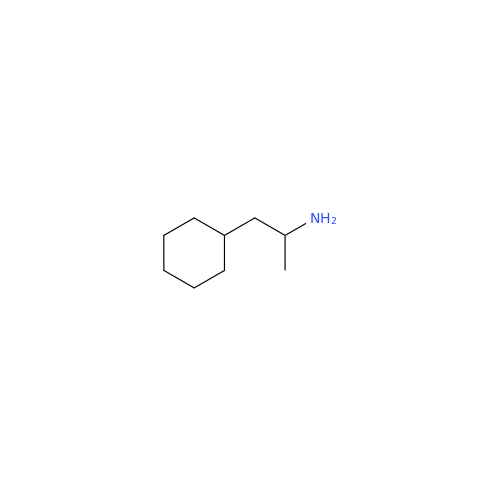
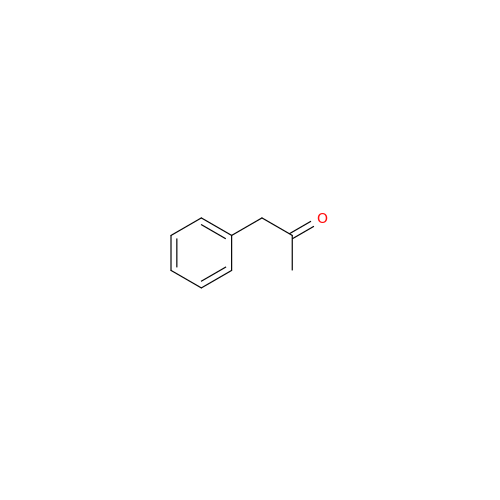
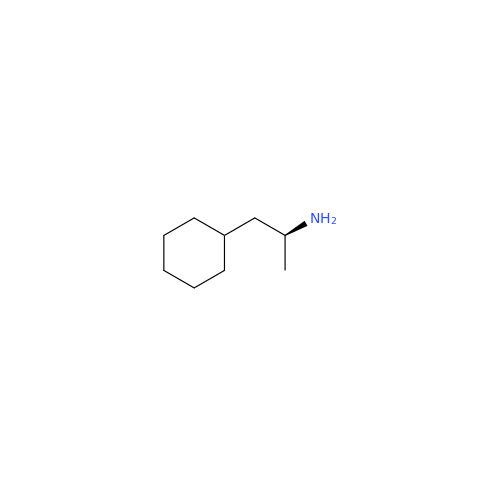
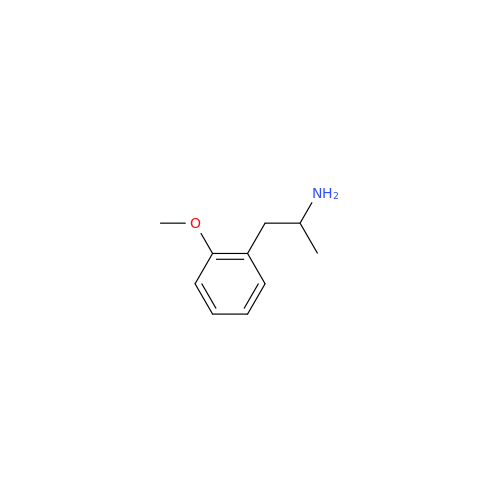
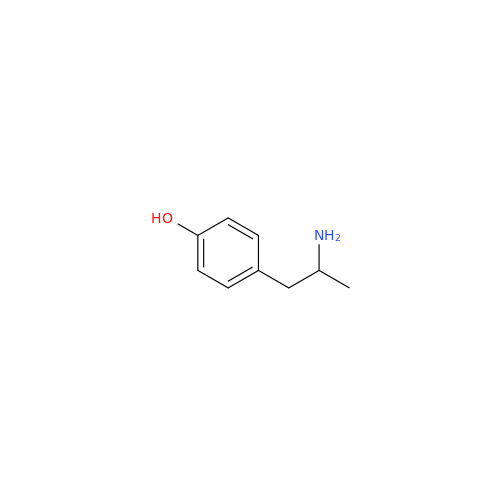
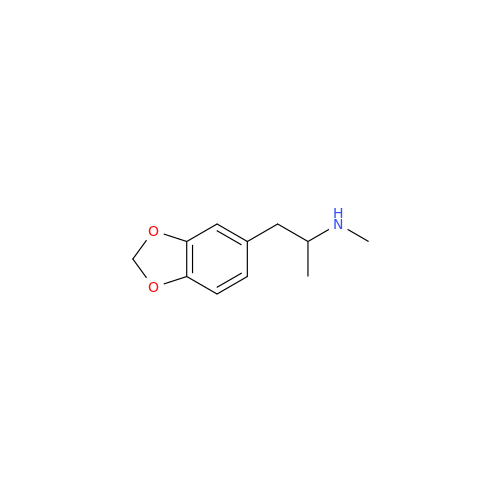
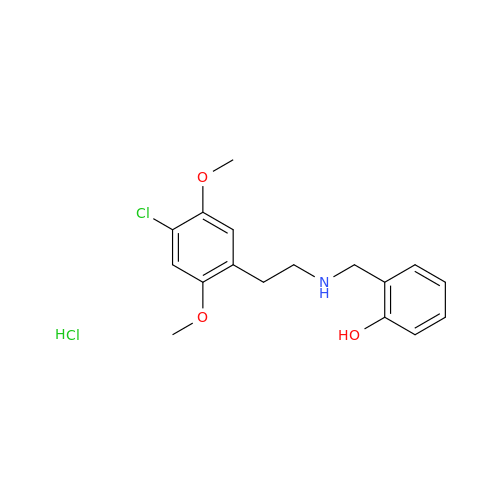
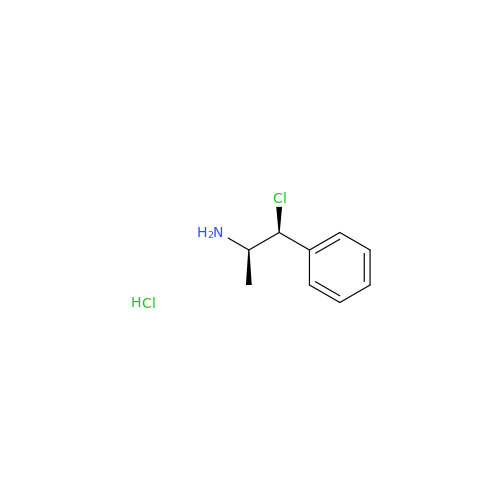
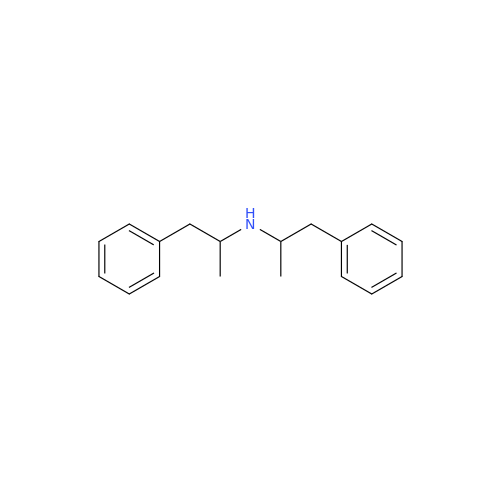
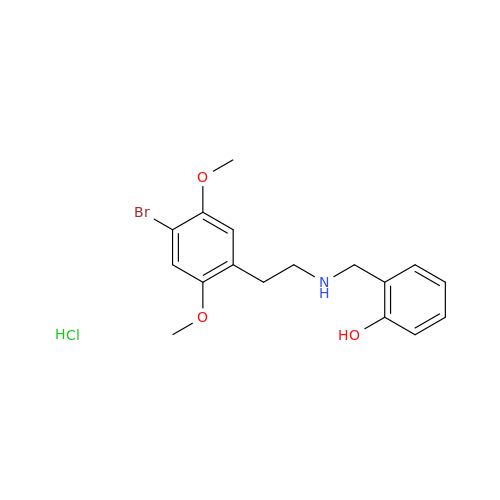
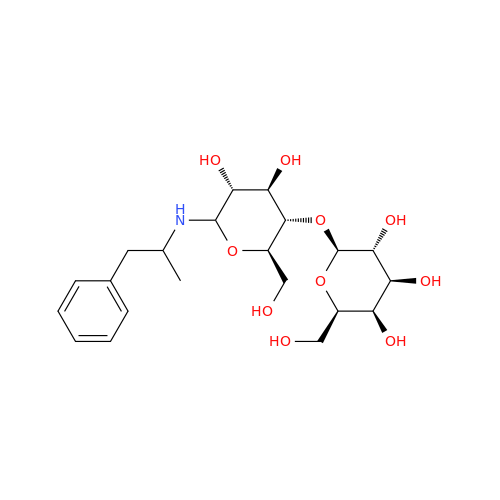
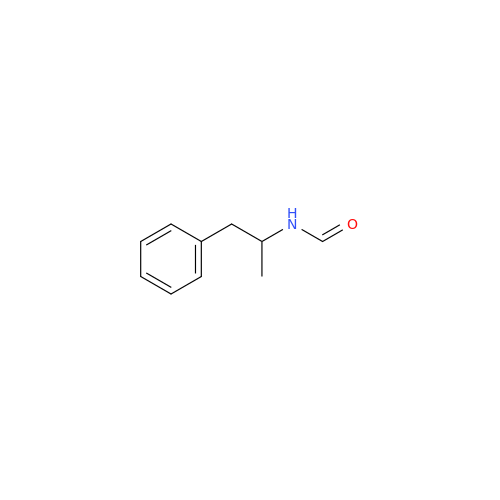
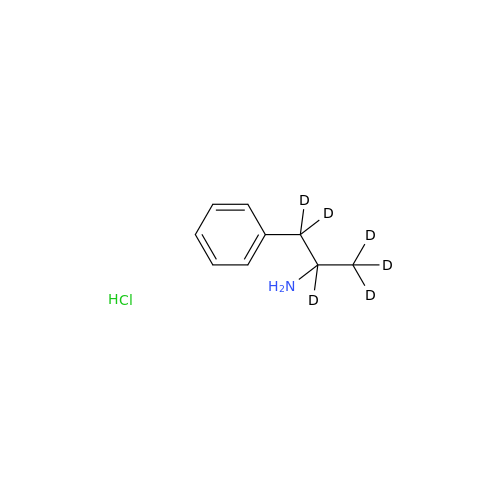
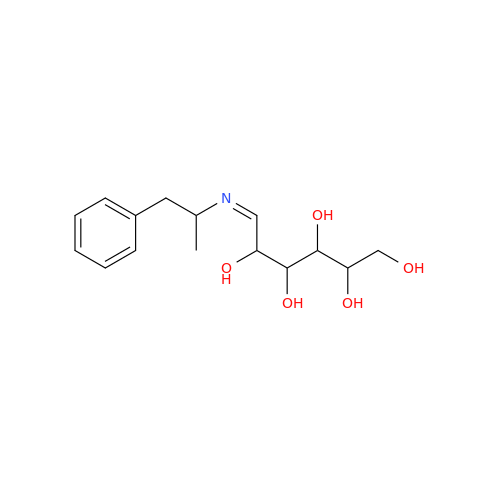
Glycosyl Amphetamine Impurity
|
Chemical Name: Glycosyl Amphetamine Impurity
Synonym: (Z)-6-((1-Phenylpropan-2-yl)imino)hexane-1,2,3,4,5-pentaol| Enter Batch Number | |||