Product Information
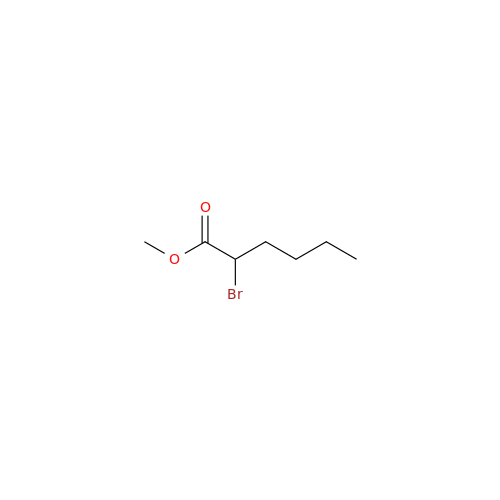
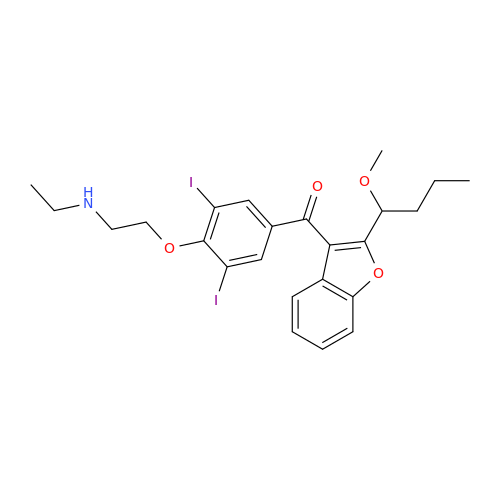
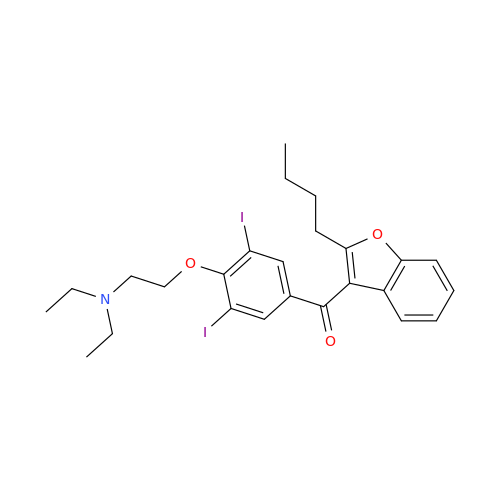
Amiodarone Impurity 19
|
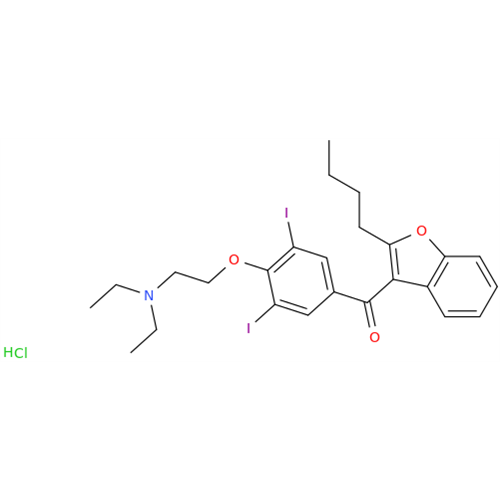
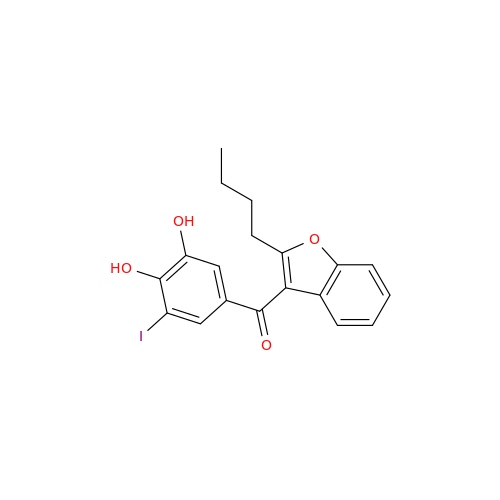
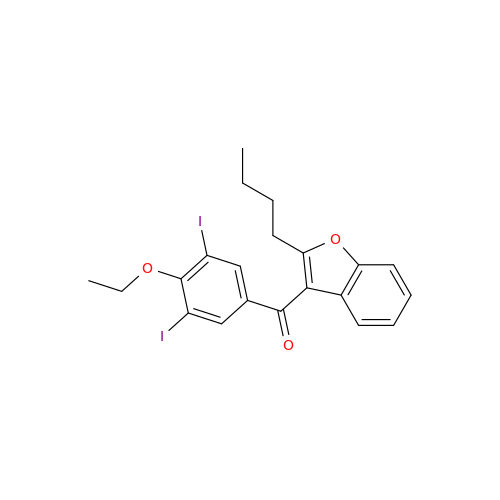
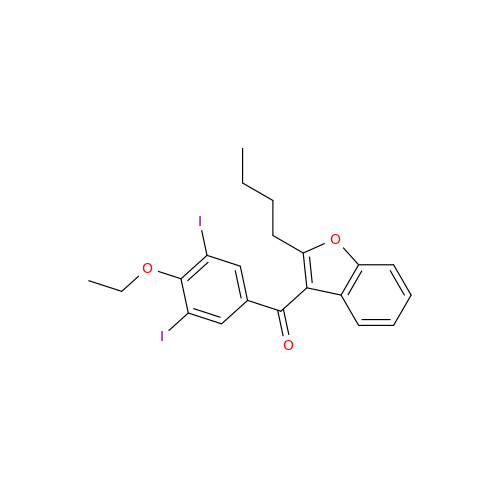
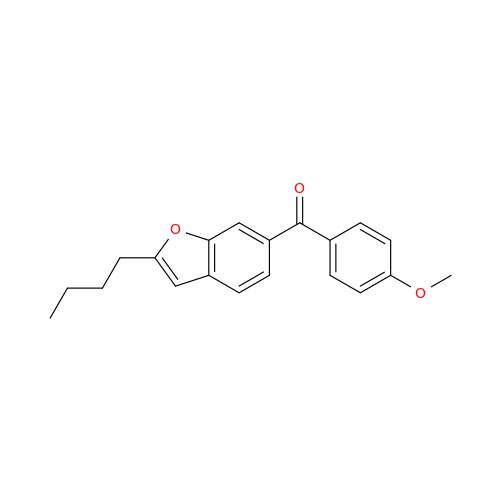
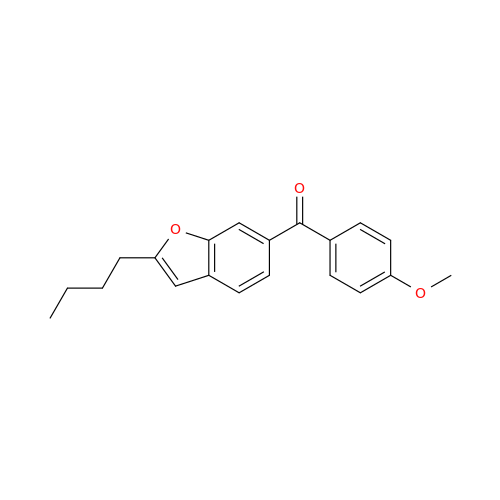
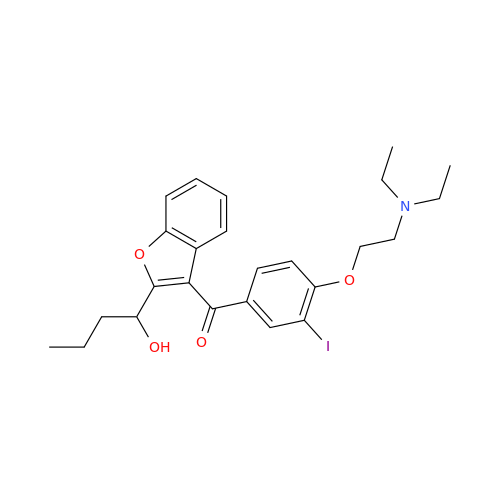
Chemical Name: Amiodarone Impurity 19
Synonym: (4-(2-(Ethylamino)ethoxy)-3,5-diiodophenyl)(2-(1-methoxybutyl)benzofuran-3-yl)methanone| Enter Batch Number | |||



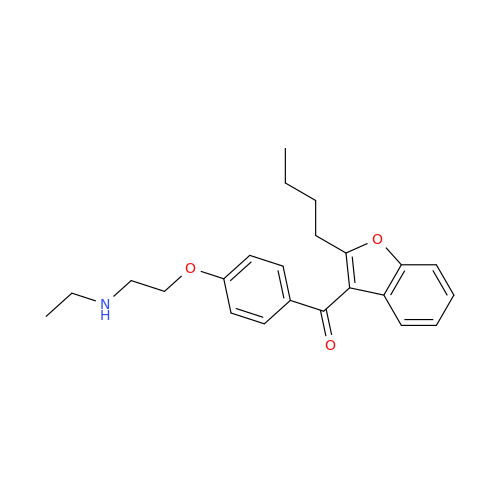
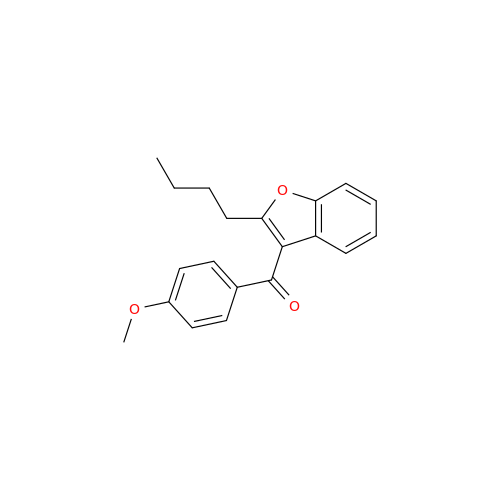
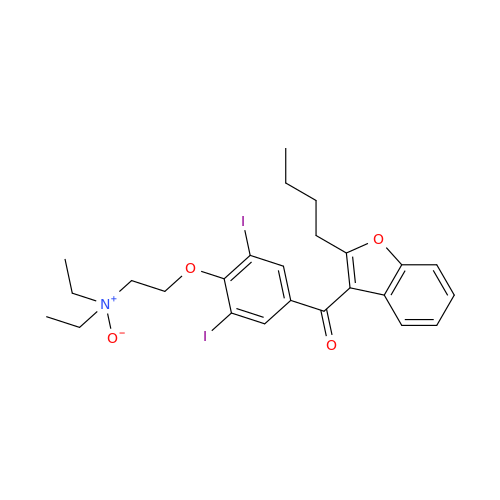
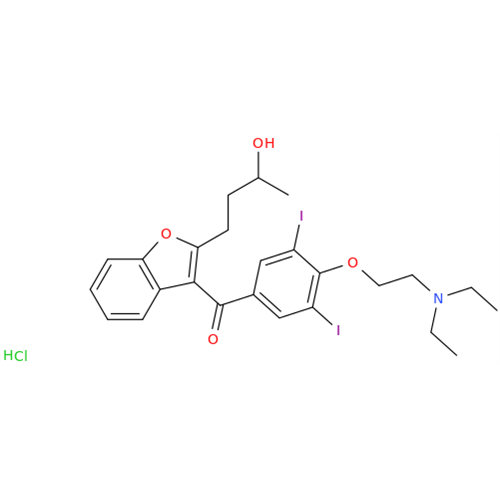
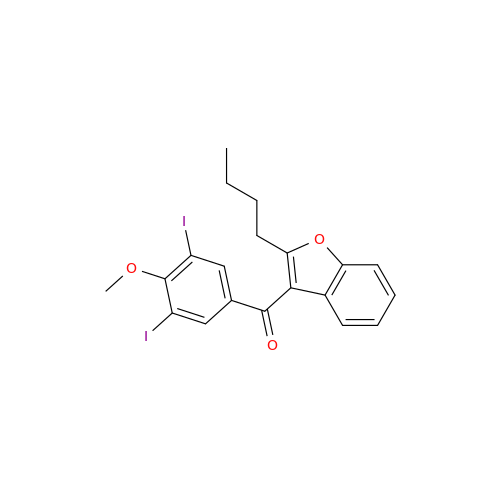
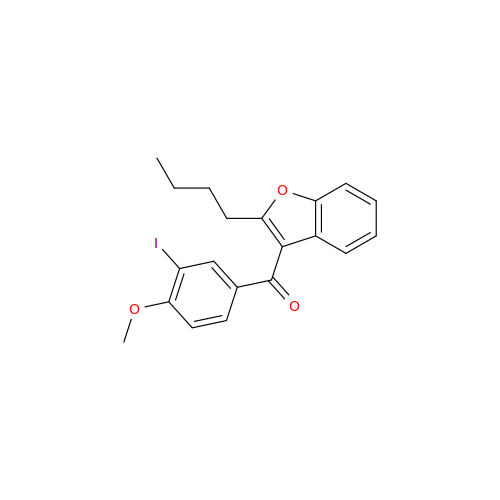
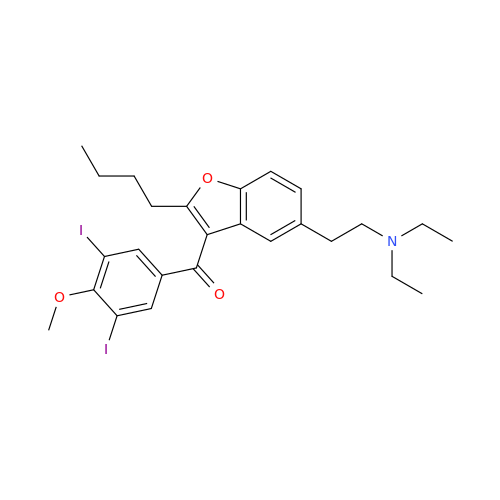
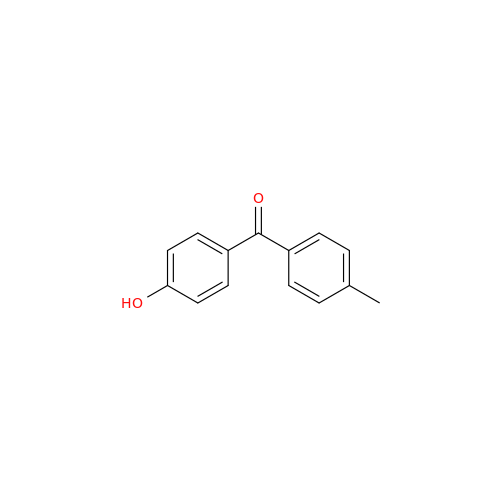
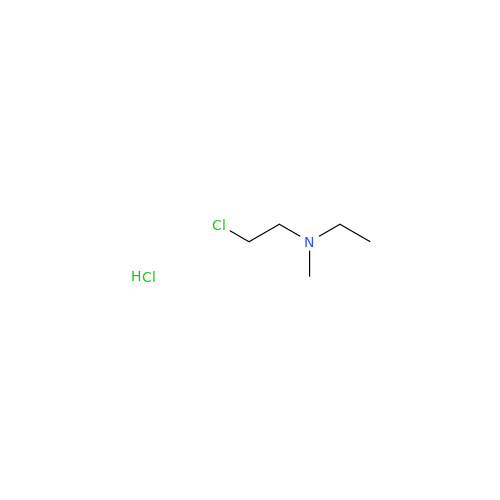
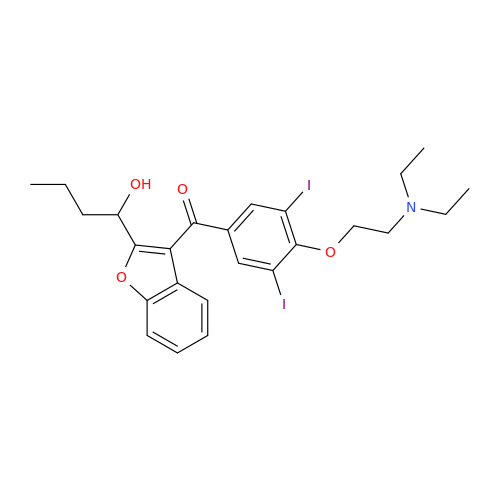
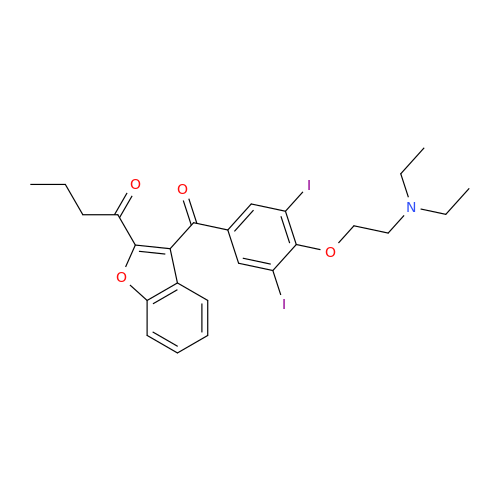
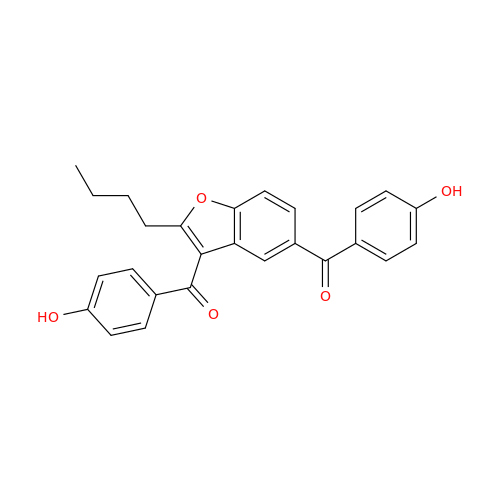
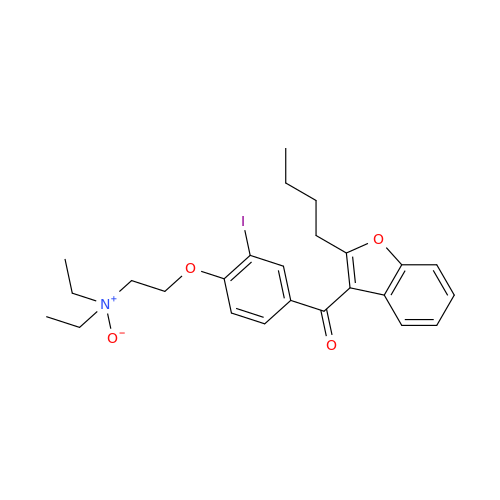
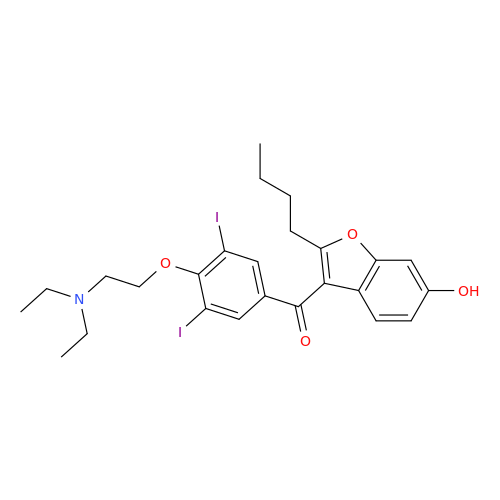
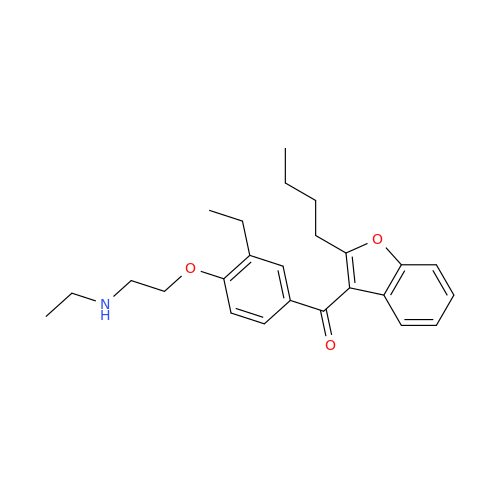
![Des-O-[2-(diethylamino)ethyl]-1-methoxy Amiodarone Des-O-[2-(diethylamino)ethyl]-1-methoxy Amiodarone](/uploads/product-details/ami040-6374.png)