Product Information
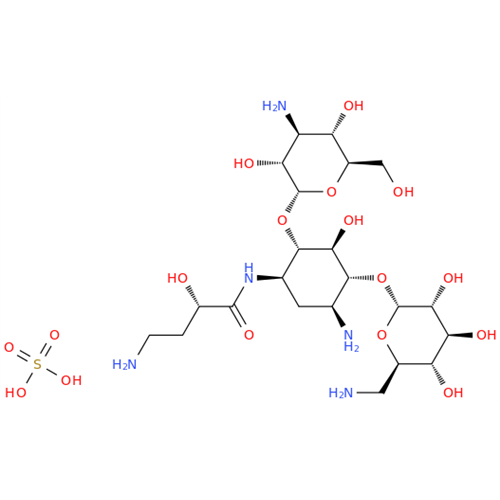
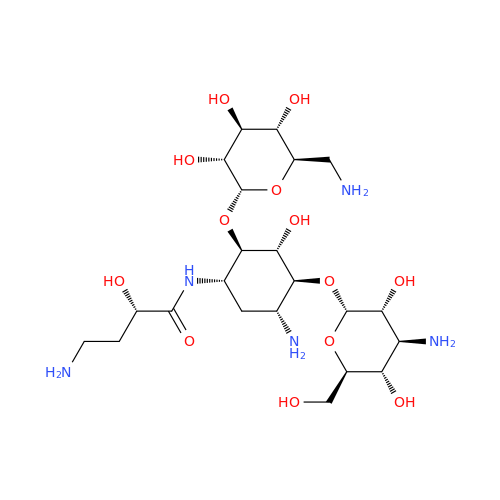
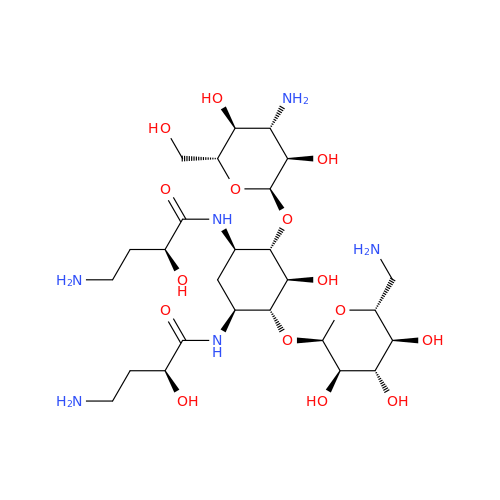
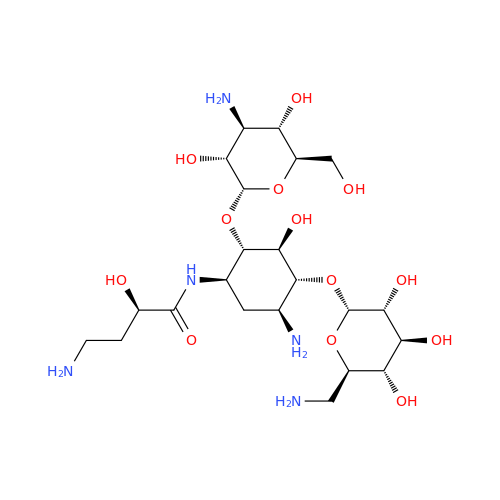
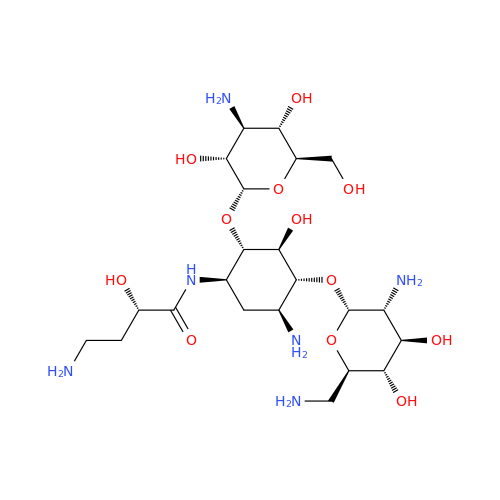
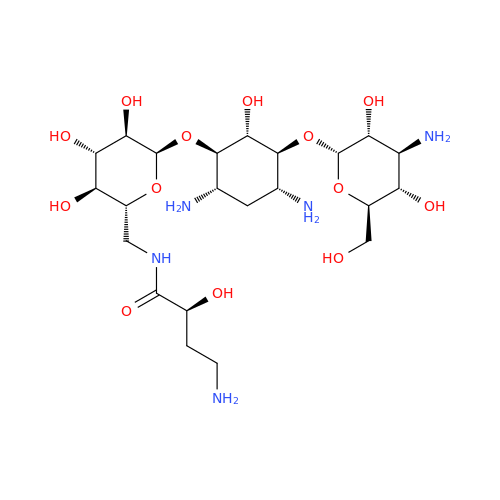
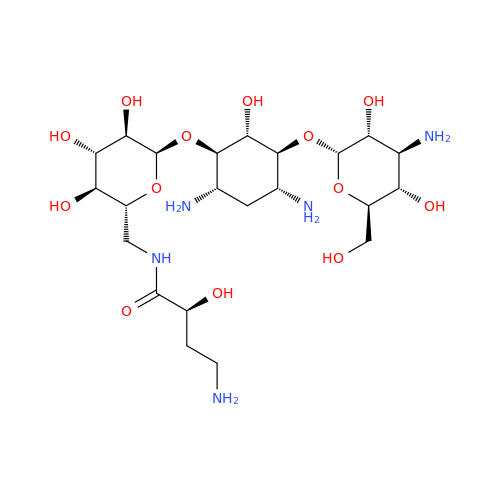
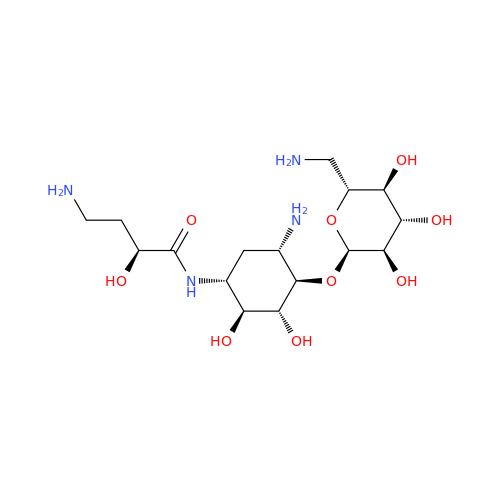
Amikacin EP Impurity D
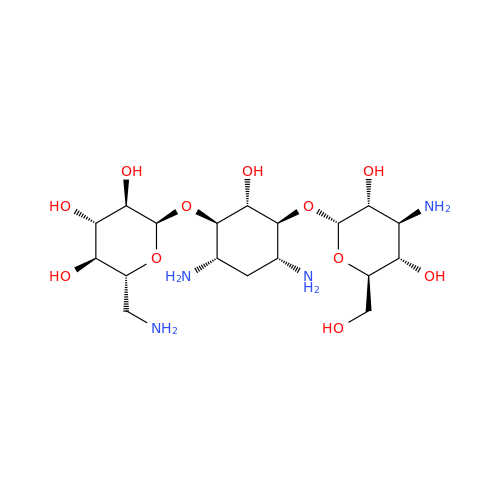
|
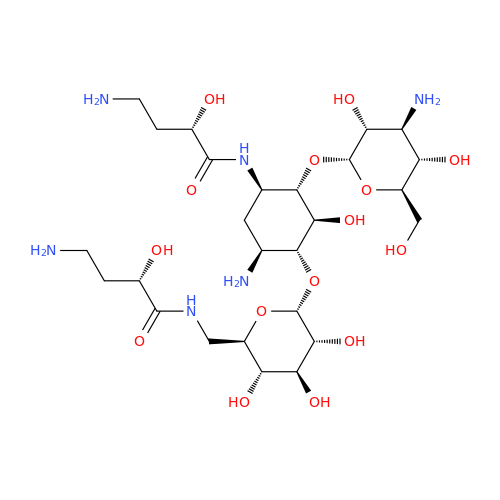
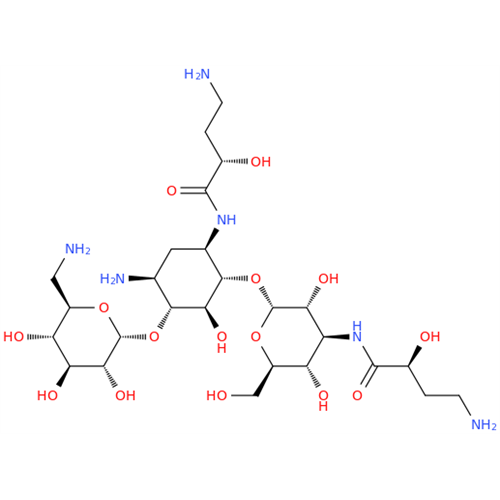
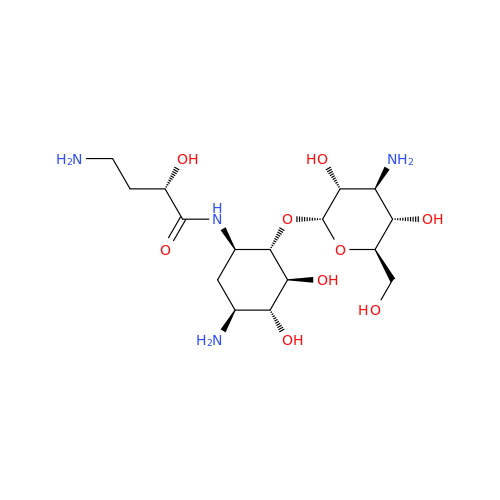
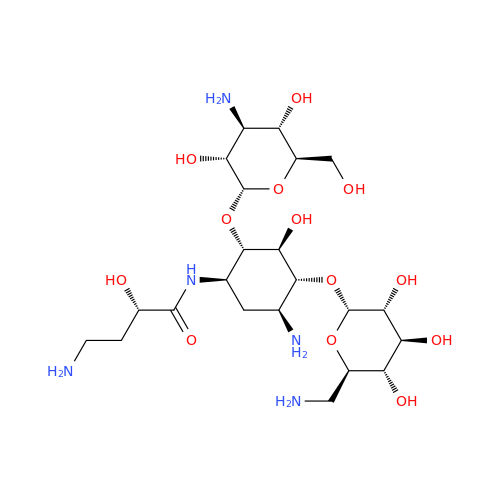
Chemical Name: Amikacin EP Impurity D
Synonym: Kanamycin API; Amikacin Sulfate Impurity D (As per 35.1 EP Monograph); (2R,3S,4S,5R,6R)-2-(Aminomethyl)-6-(((1R,2R,3S,4R,6S)-4,6-diamino-3-(((2S,3R,4S,5S,6R)-4-amino-3,5-dihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)oxy)-2-hydroxycyclohexyl)oxy)tetrahydro-2H-pyran-3,4,5-triol;6-O-(3-Amino-3-deoxy-α-D-glucopyranosyl)-4-O-(6-amino-6-deoxy-α-D-glucopyranosyl)-2-deoxy-D-streptamine| Enter Batch Number | |||