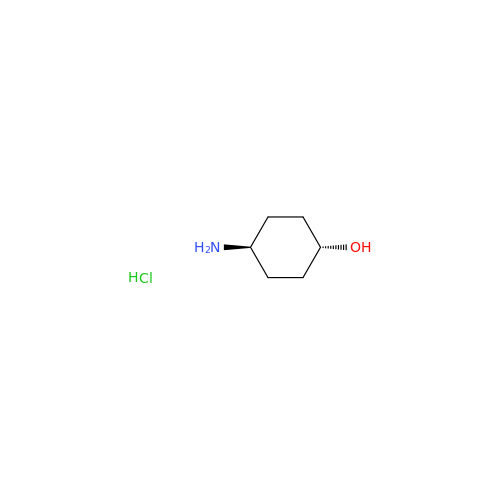
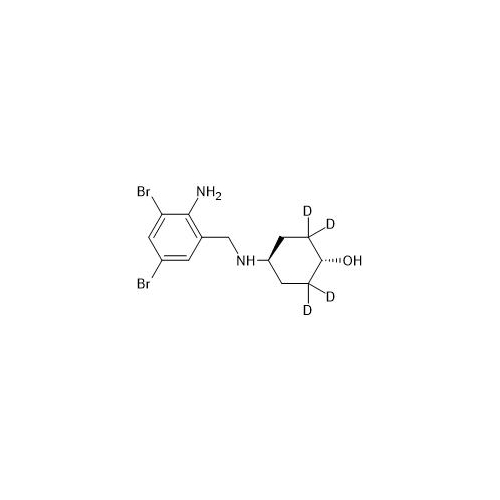
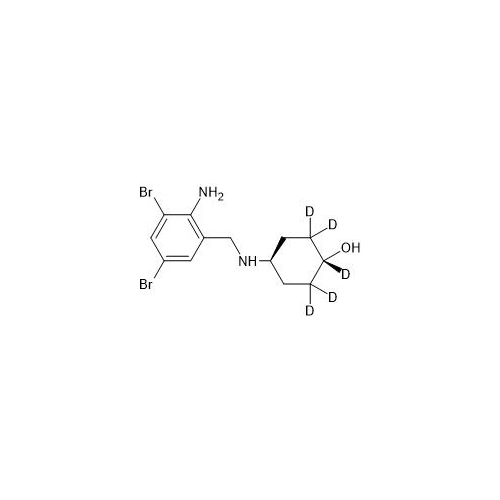
Product Information
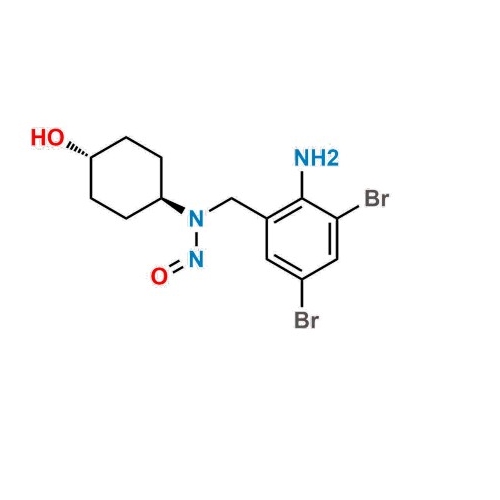
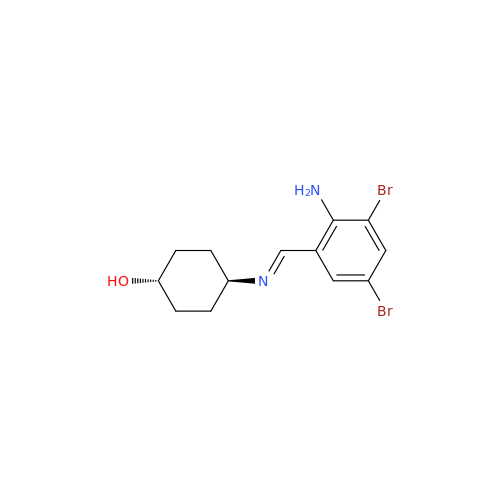
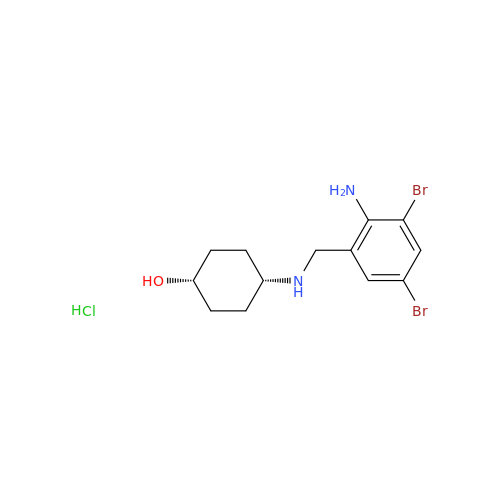
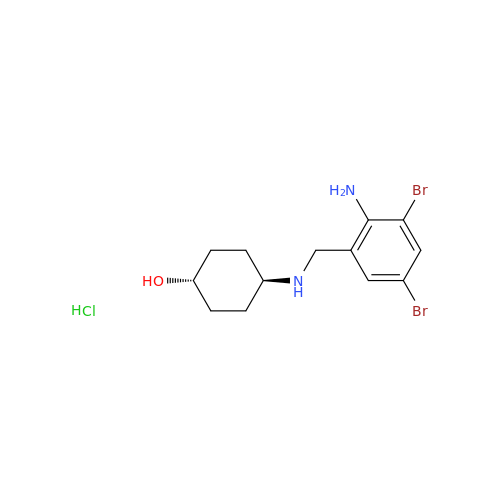
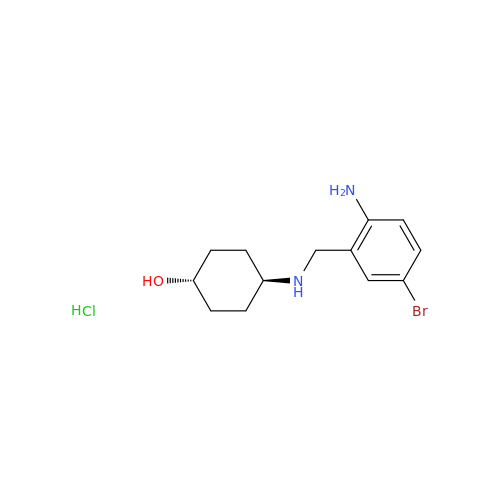
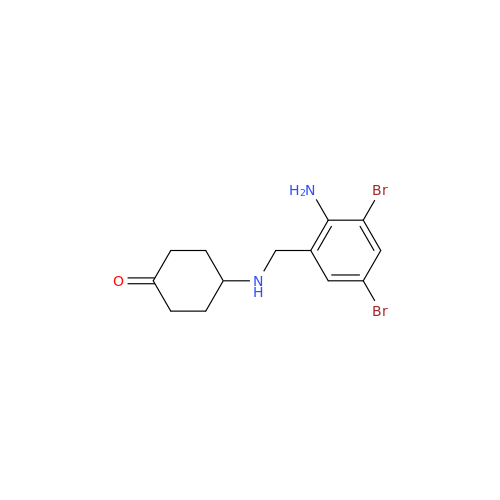
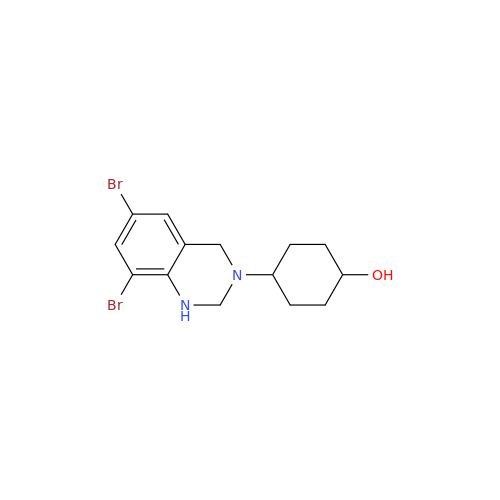
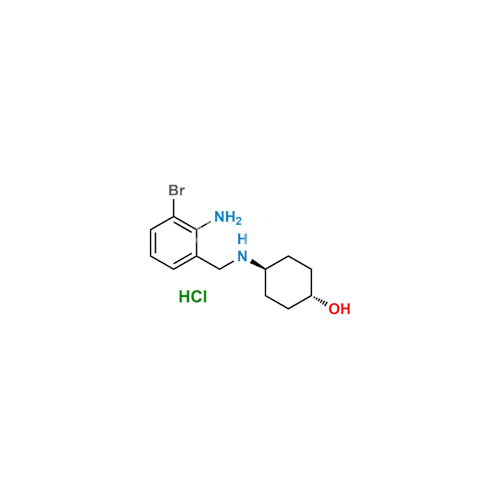
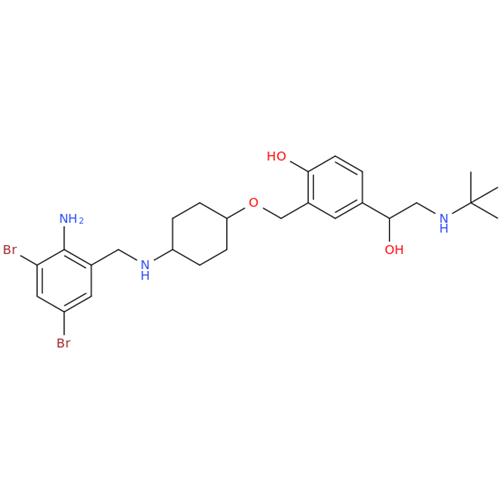
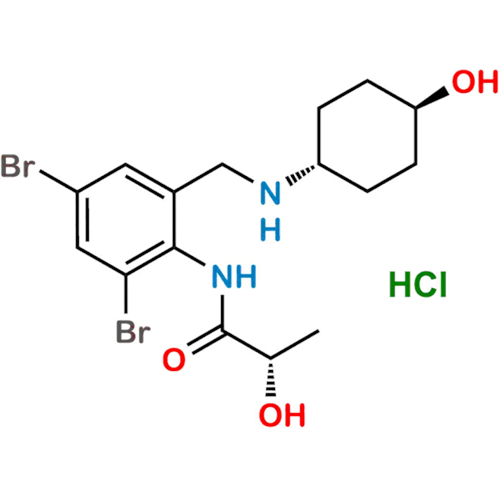
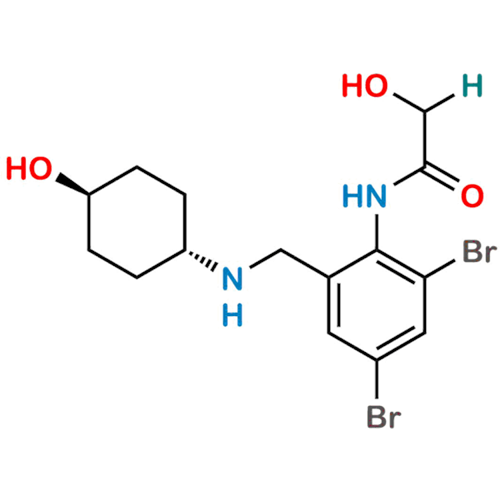
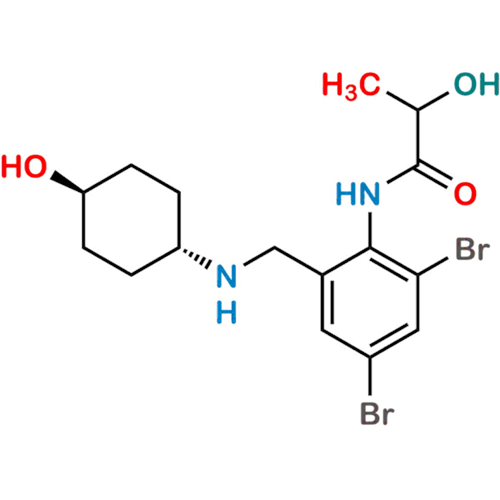
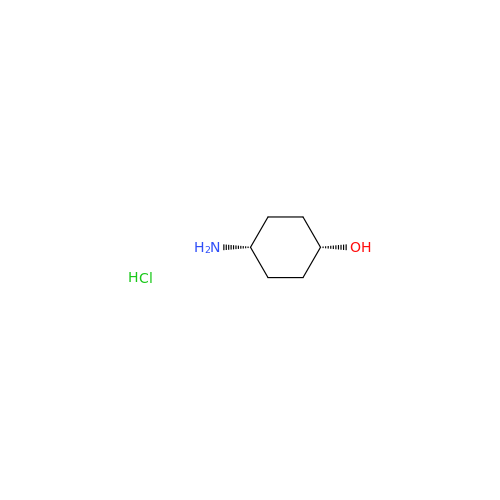
Ambroxol Impurity 3
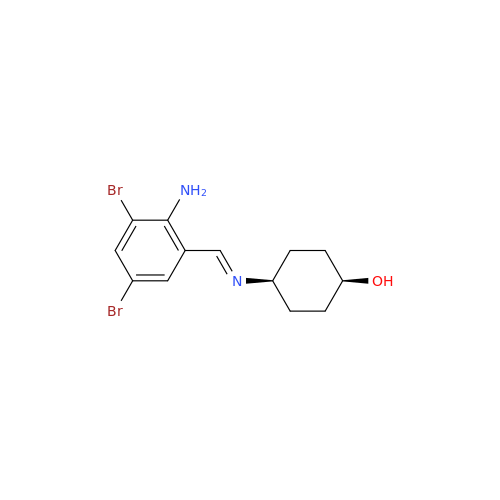
|
Chemical Name: Ambroxol Impurity 3
Synonym: Cyclohexanol, 4-[[(2-amino-3,5-dibromophenyl)methylene]amino]-, cis; rel-(1s,4s)-4-((2-amino-3,5-dibromobenzylidene)amino)cyclohexan-1-ol| Enter Batch Number | |||