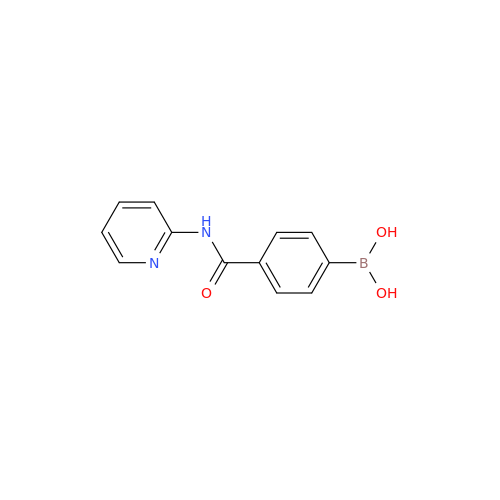
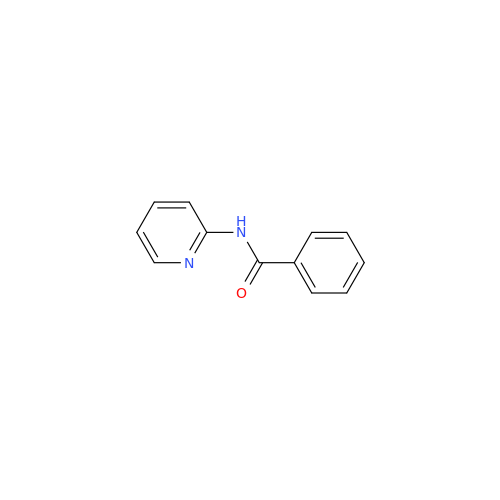
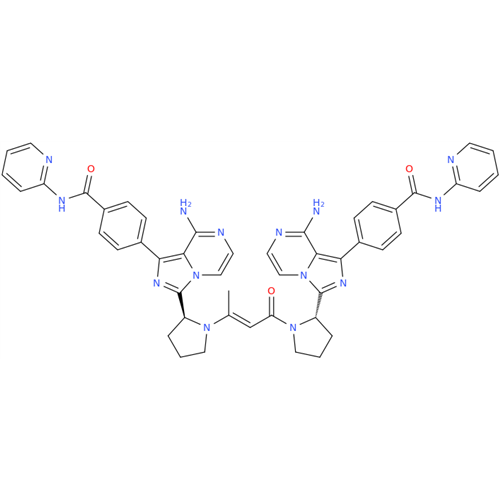
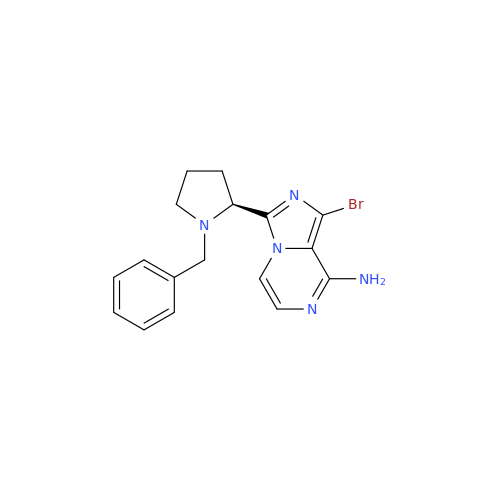
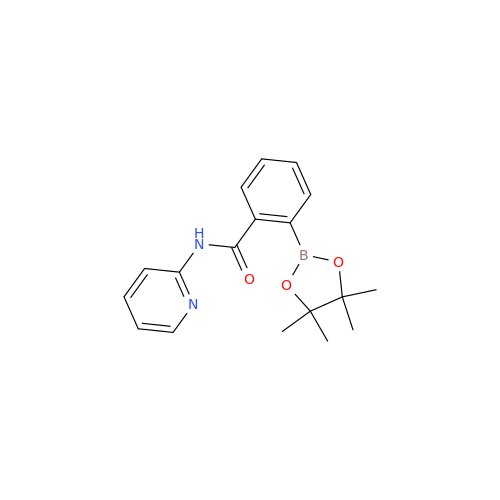
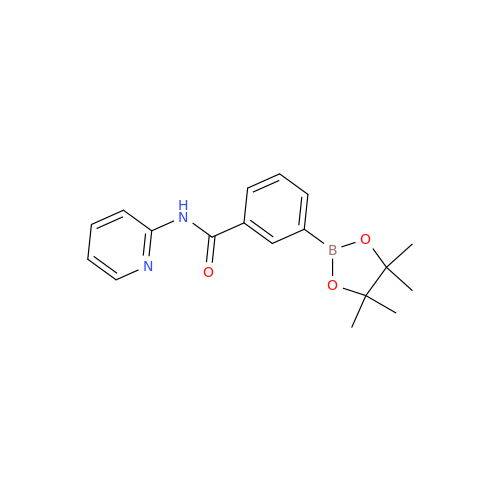
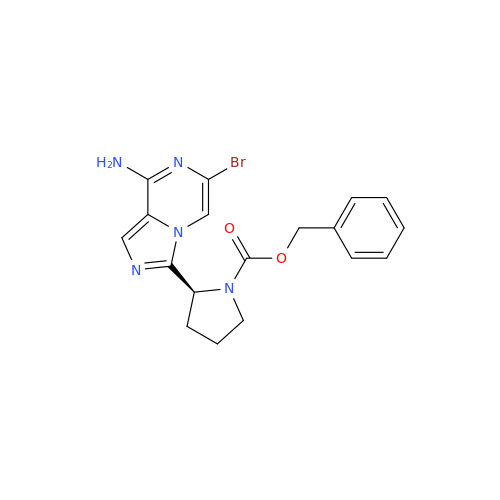
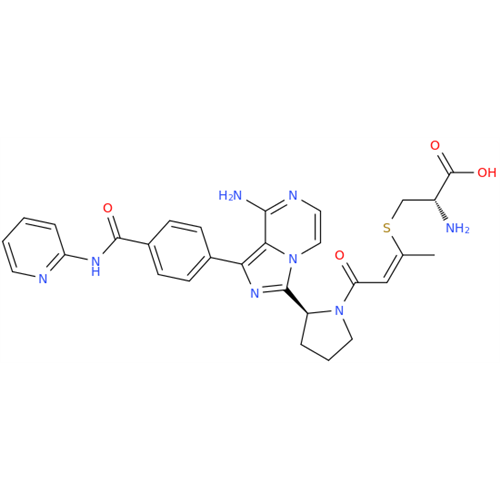
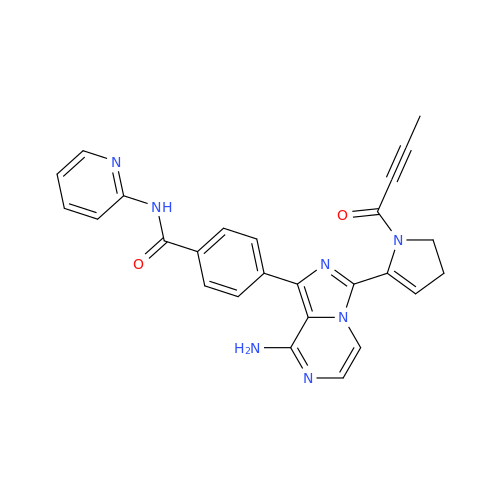
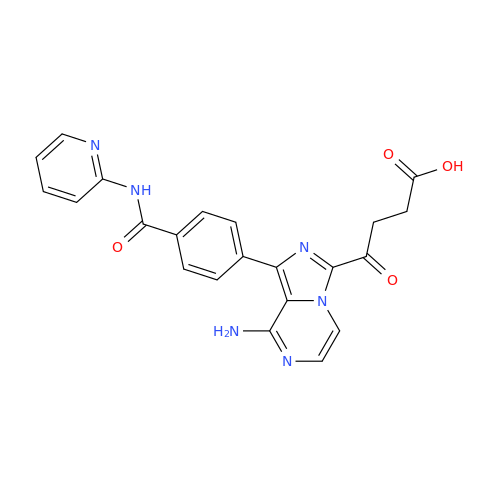
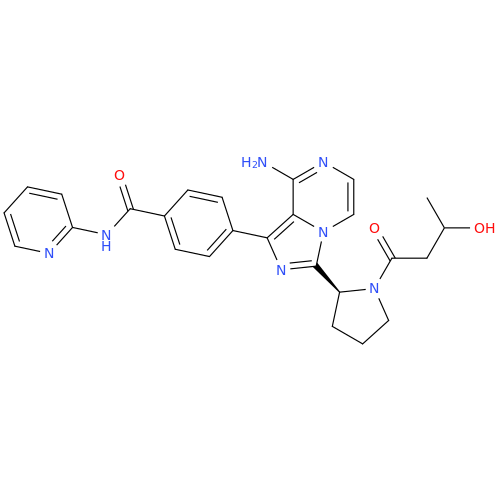
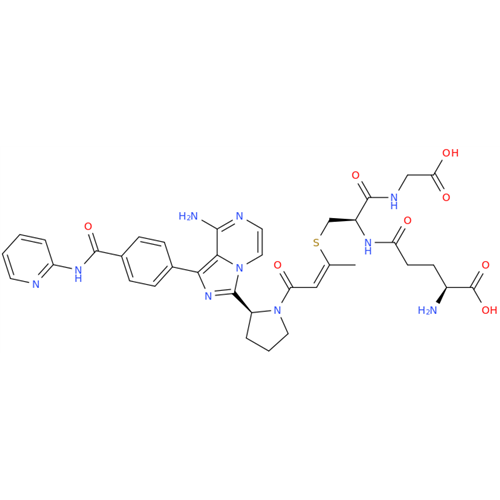
Product Information
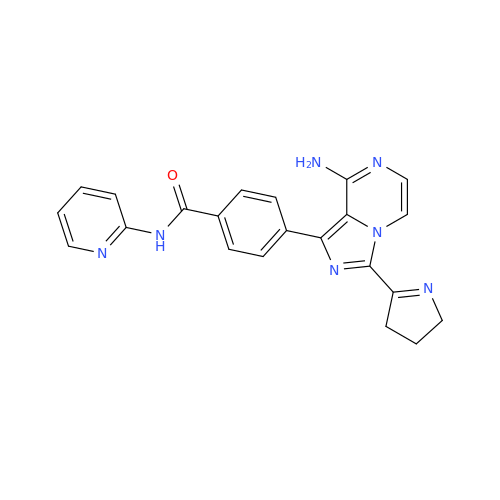
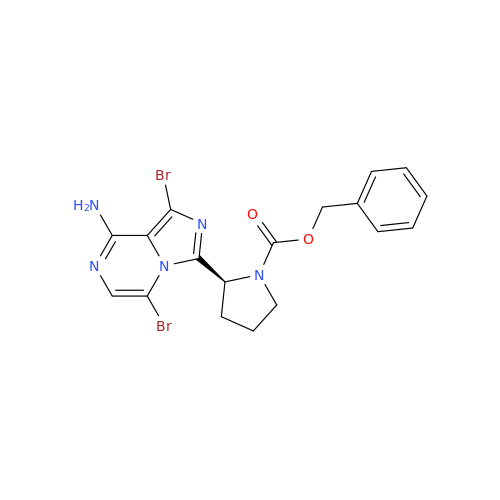
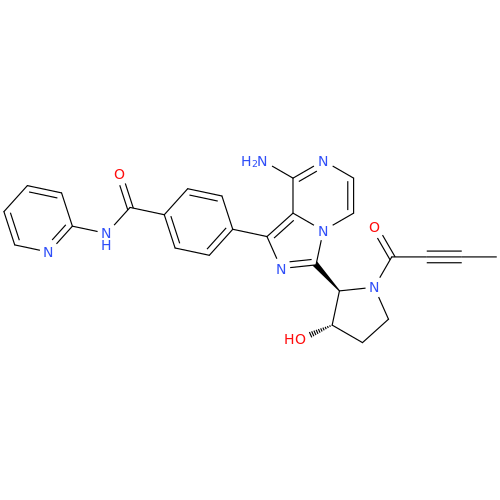
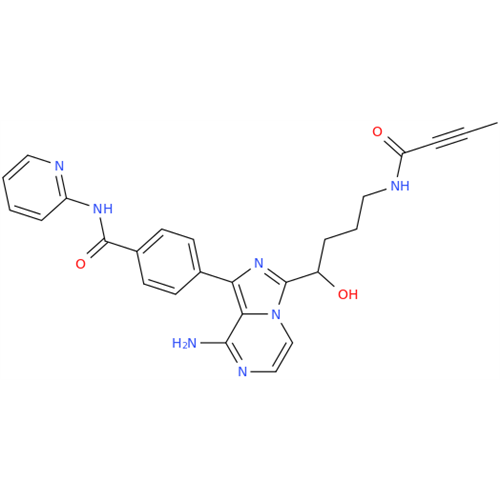
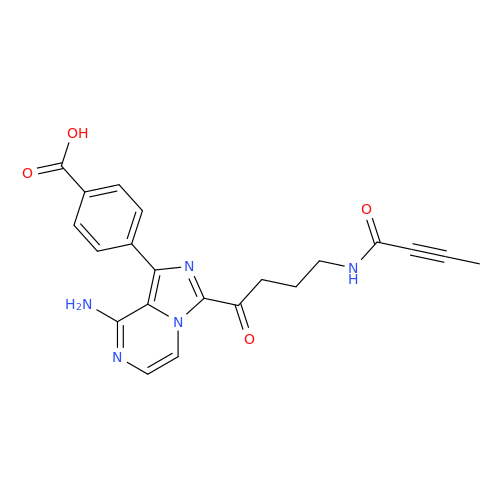
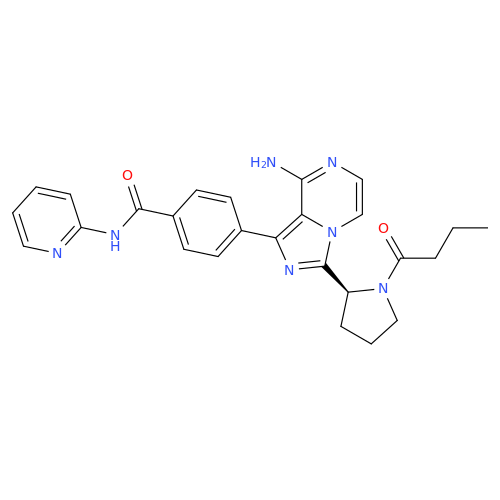
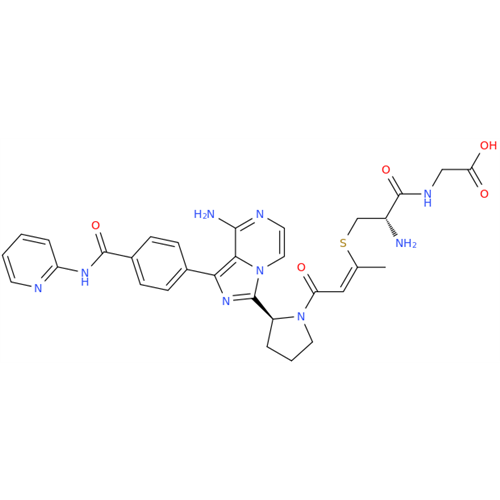
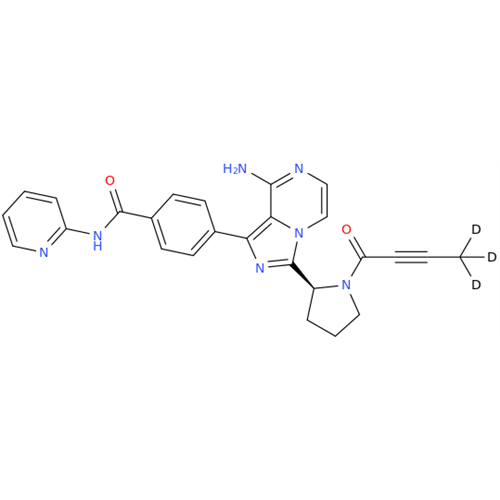
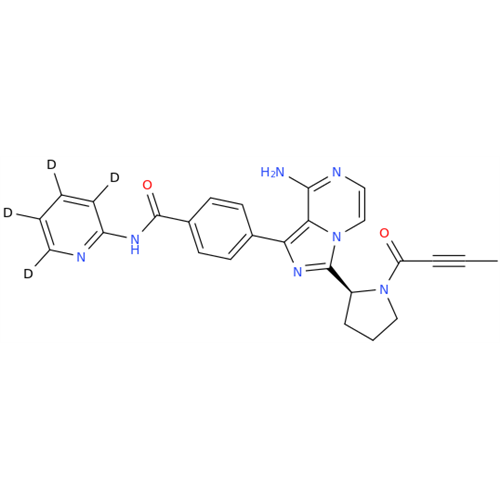
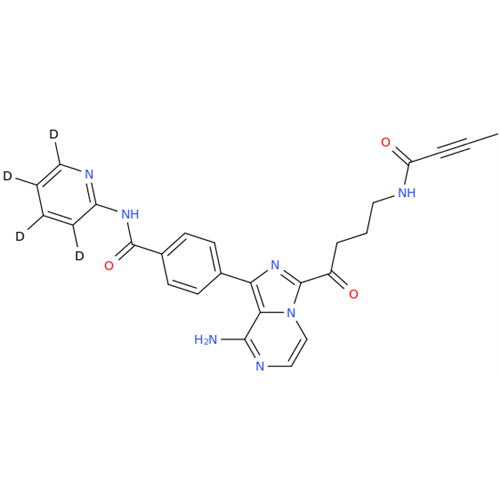
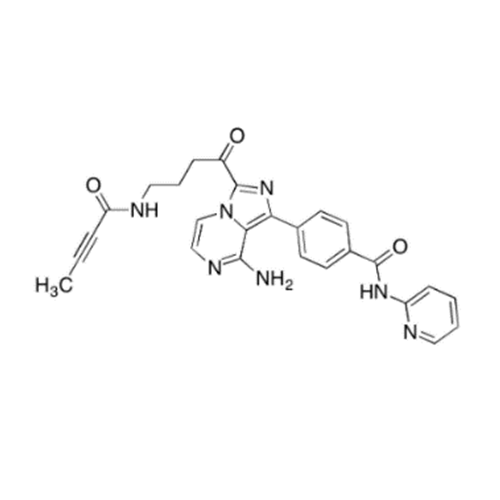
Acalabrutinib Impurity 35
|
Chemical Name: Acalabrutinib Impurity 35
Synonym: Acalabrutinib Impurity 35| Enter Batch Number | |||



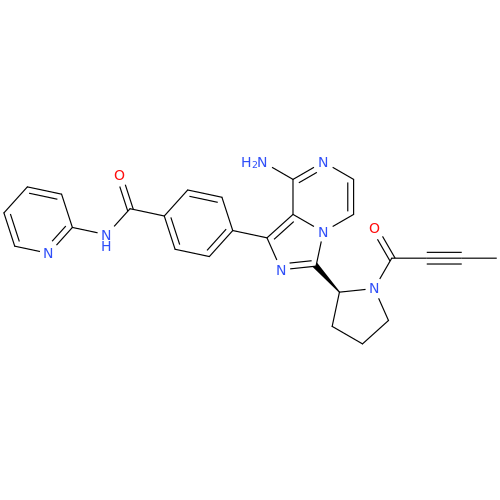
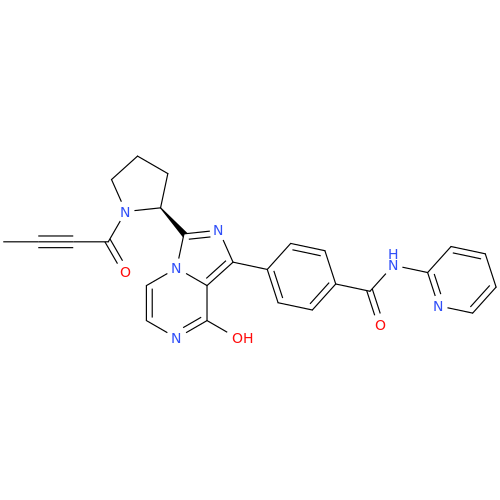
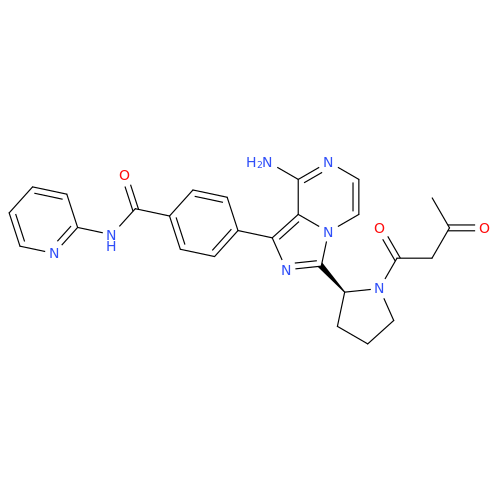
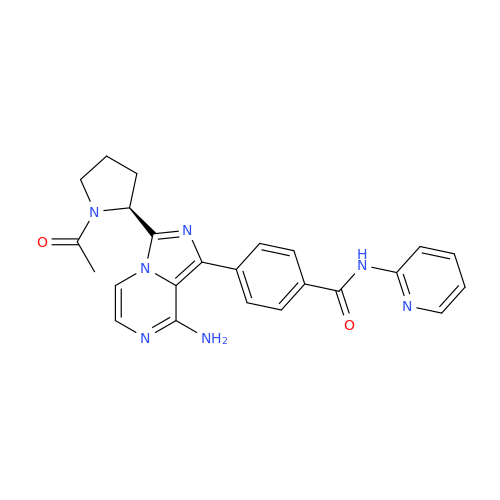
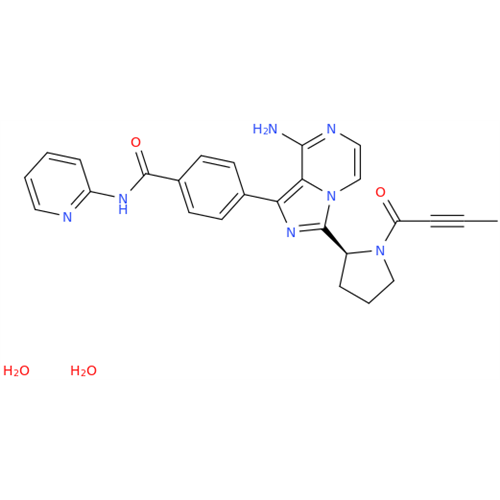
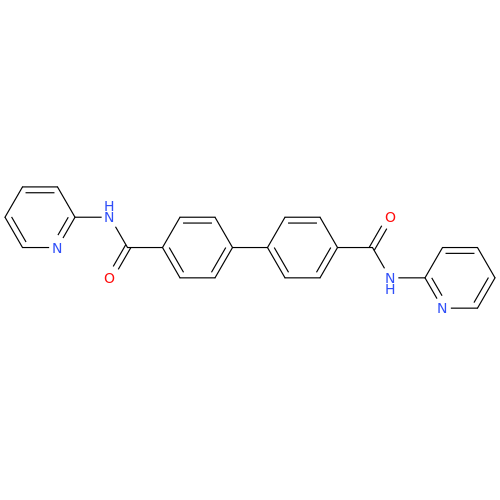
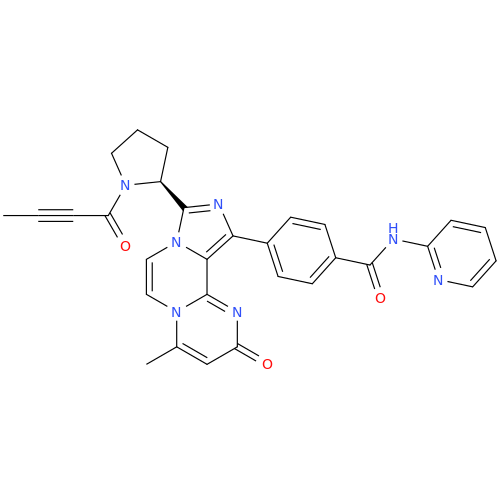
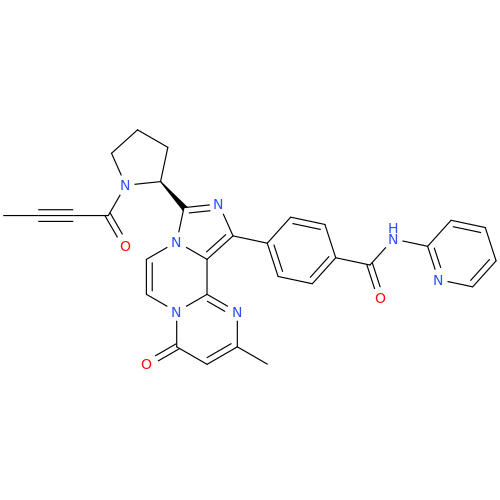
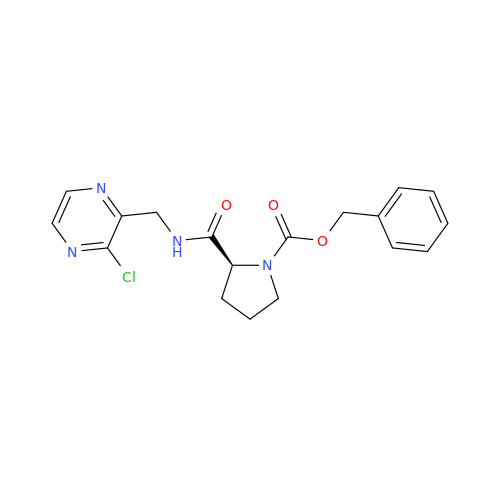
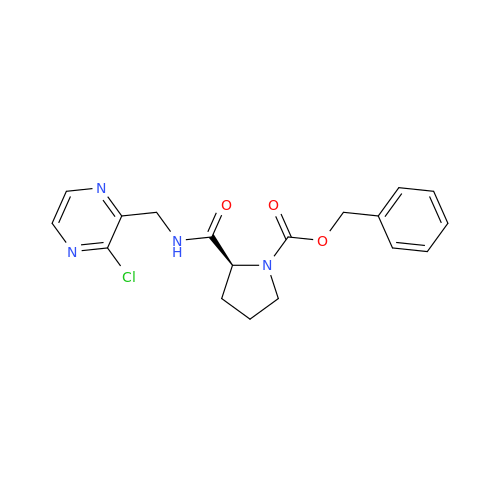
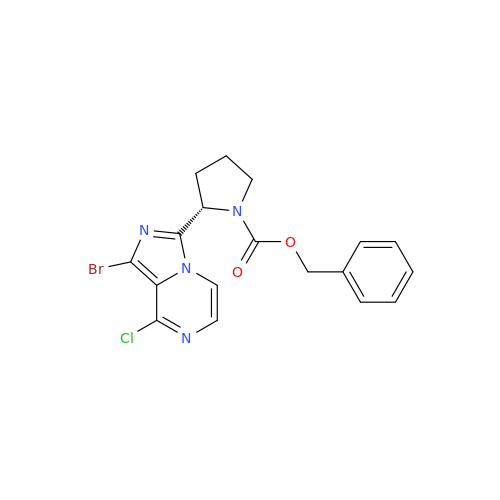
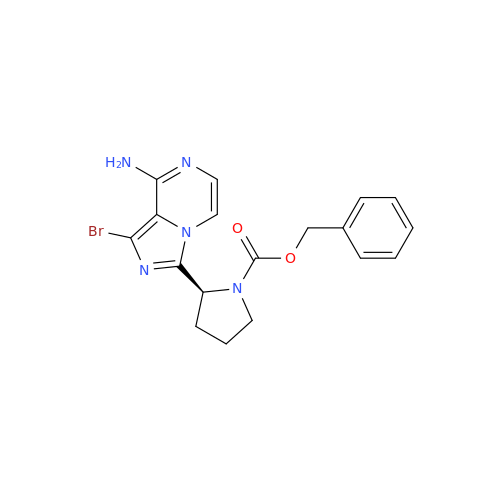
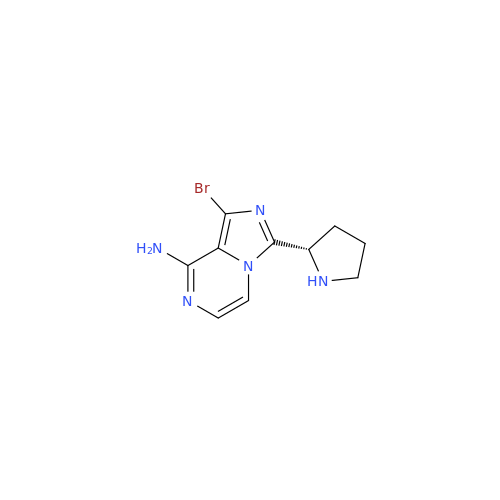
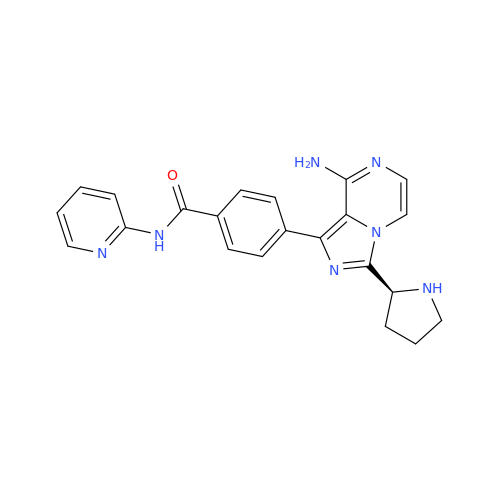
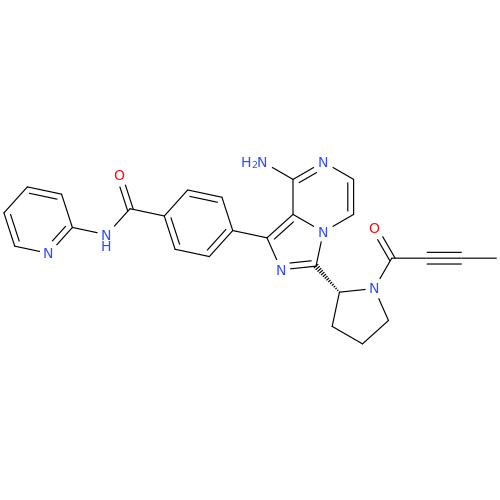
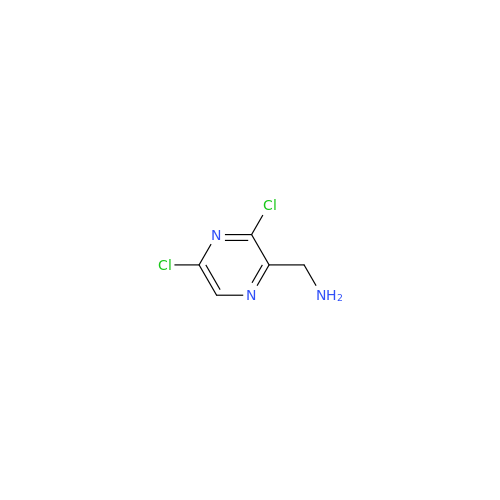
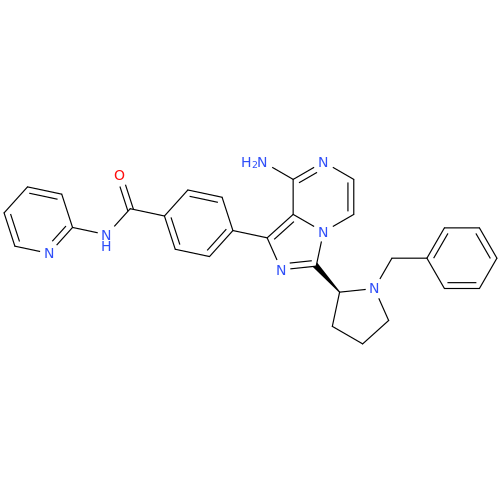
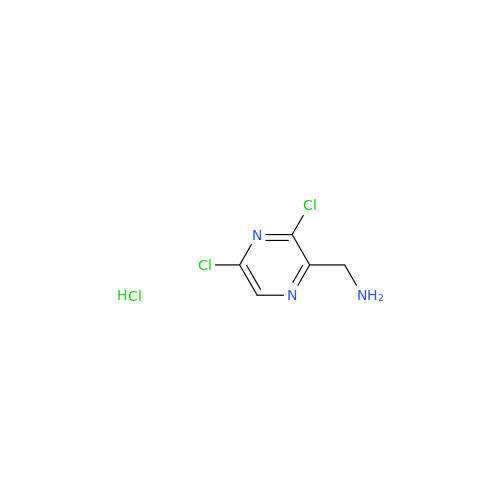
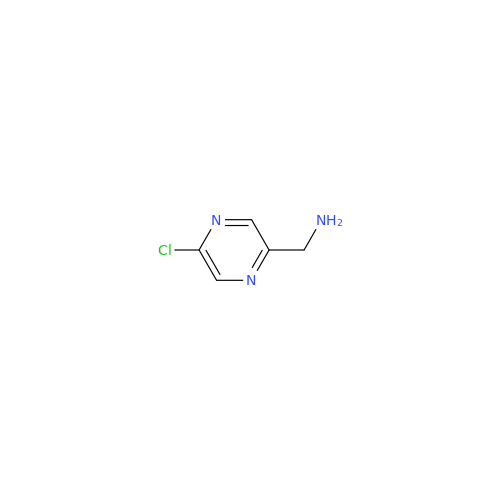
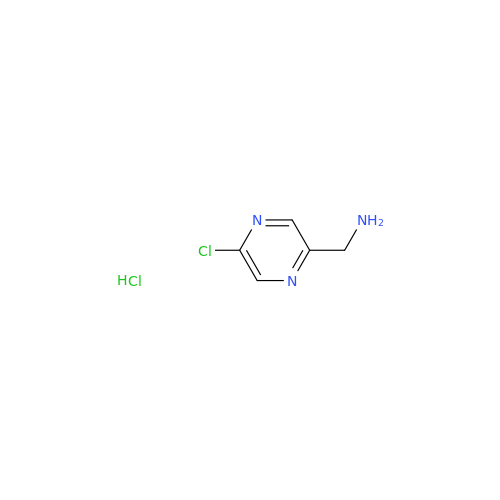
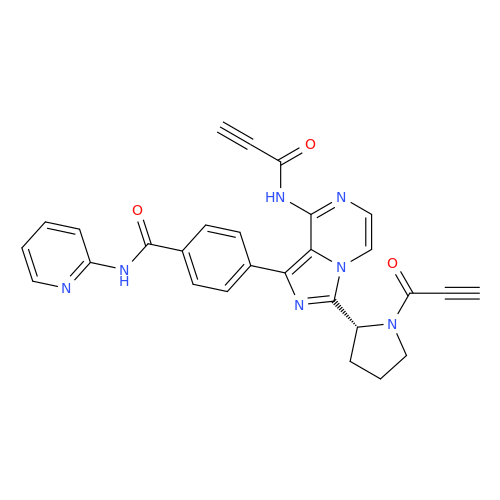
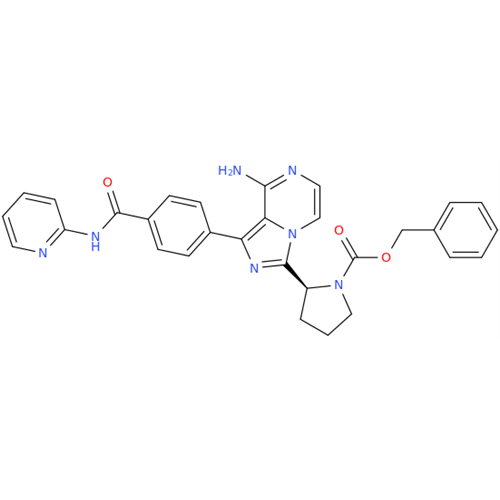
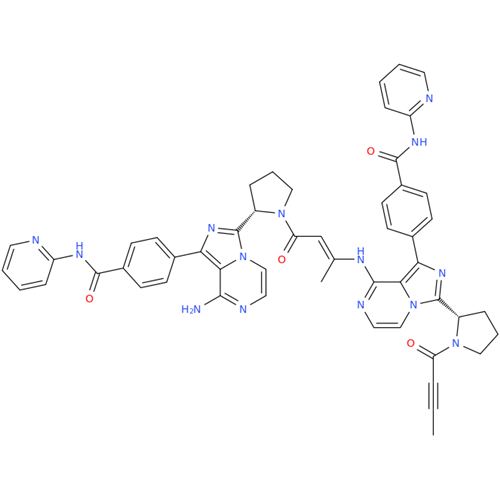
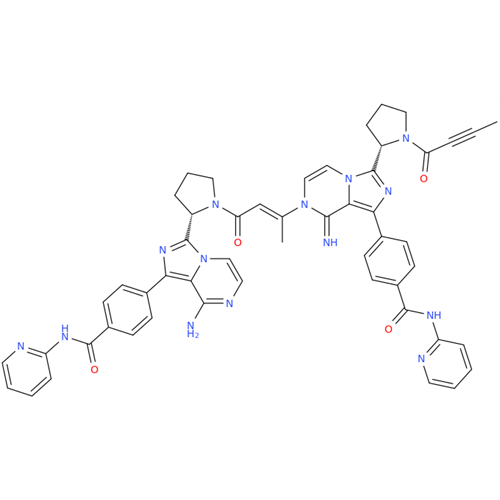
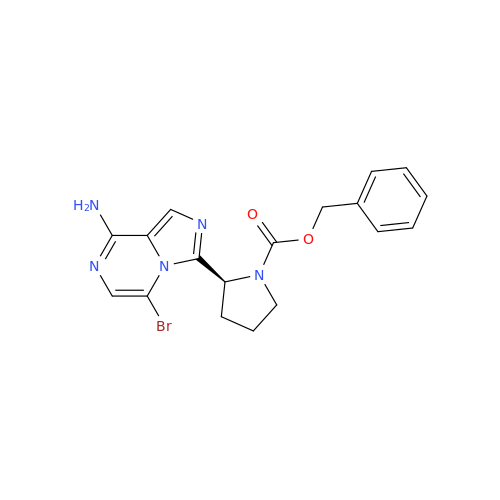
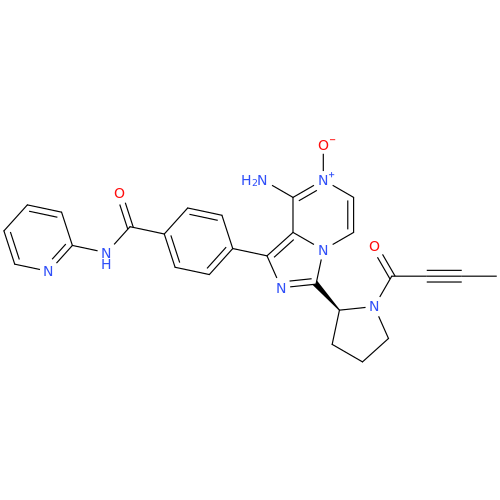
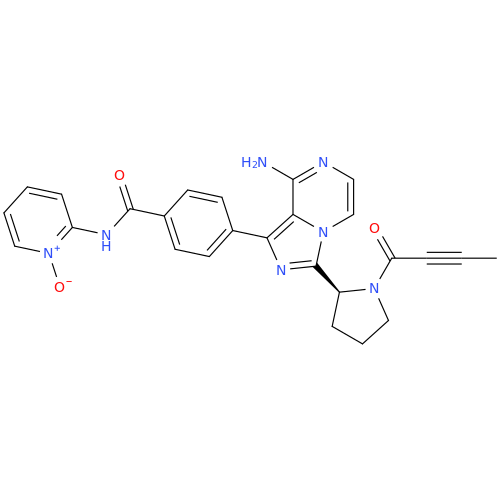
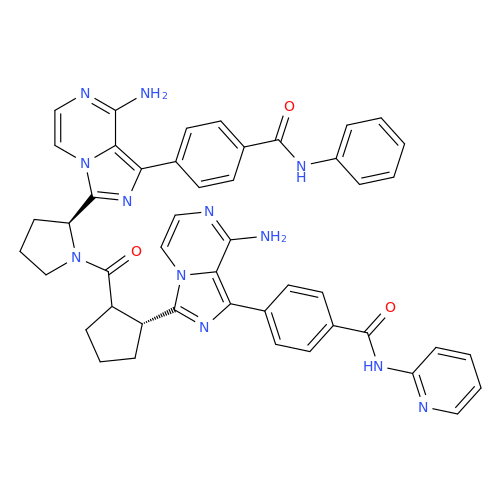
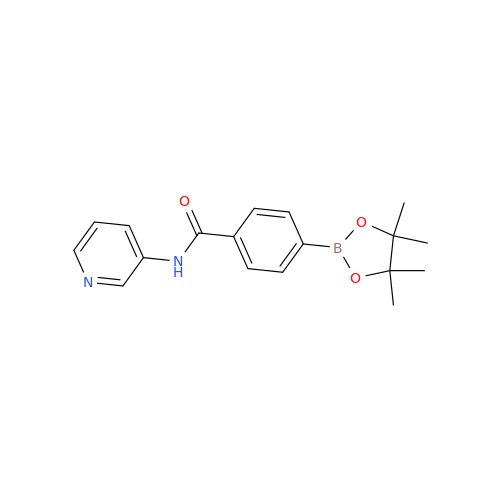
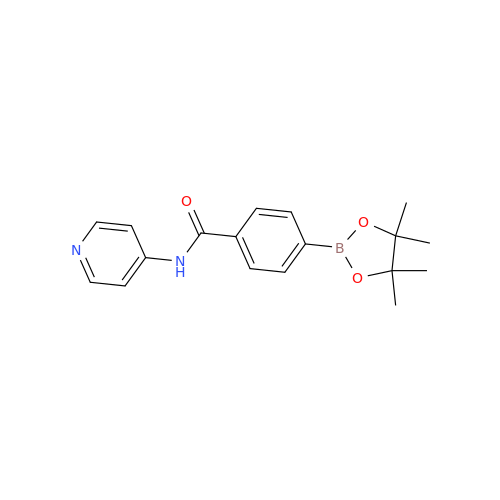
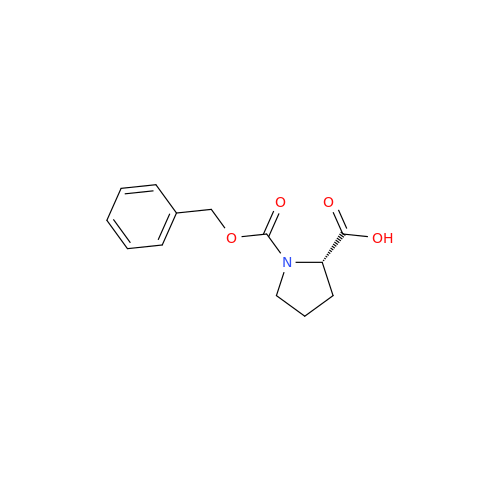
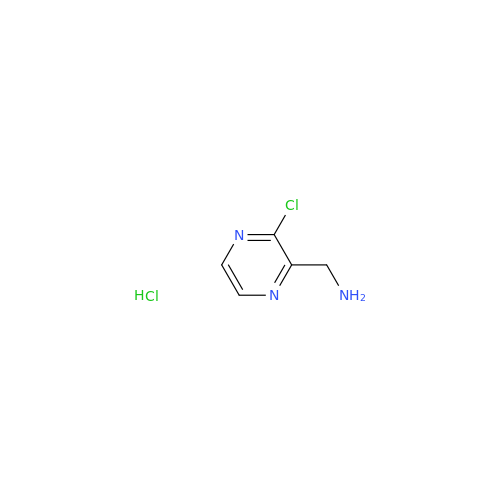
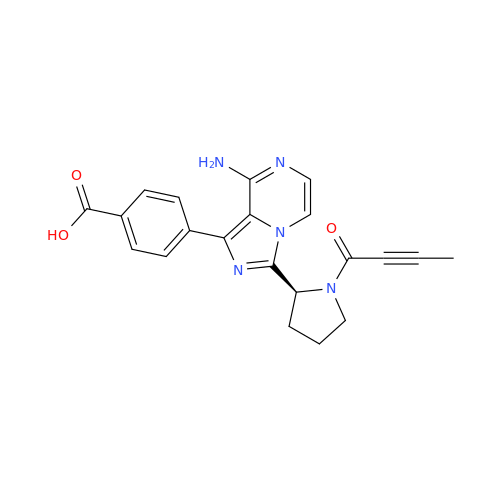
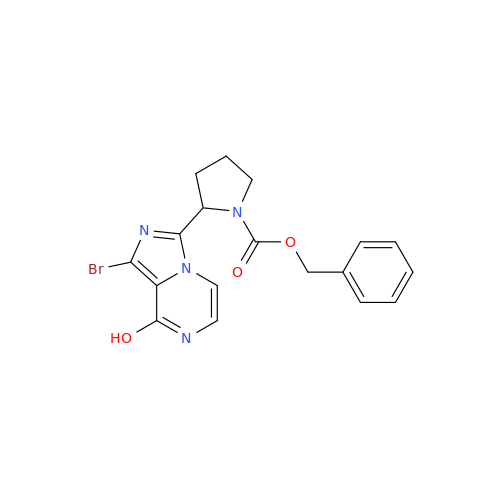
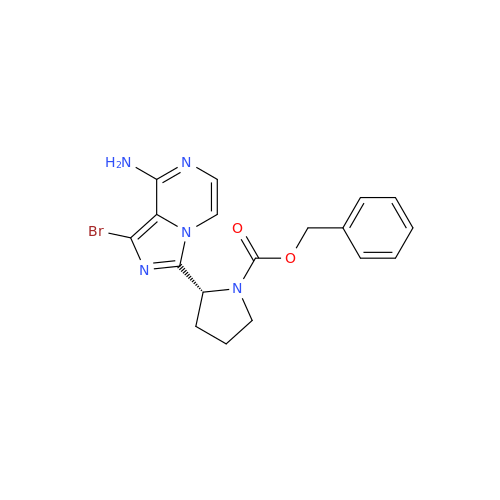
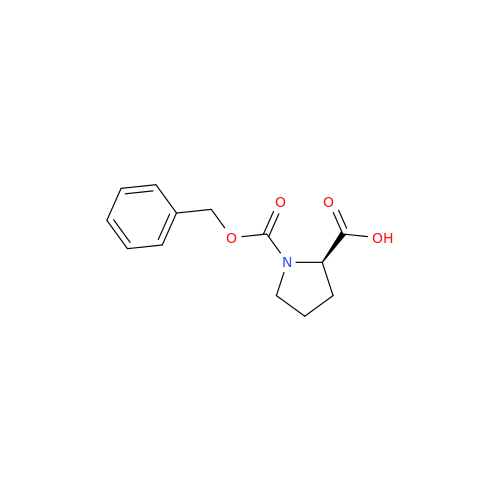
![Benzyl (S)-2-(8-aminoimidazo[1,5-a]pyrazin-3-yl)pyrrolidine-1-carboxylate Benzyl (S)-2-(8-aminoimidazo[1,5-a]pyrazin-3-yl)pyrrolidine-1-carboxylate](/uploads/product-details/atb038-5030.png)